WikiJournal of Medicine/Impact of xenogenic mesenchymal stem cells secretome on a humoral component of the immune system
WikiJournal of Medicine
Open access • Publication charge free • Public peer review • Wikipedia-integrated
This article has been through public peer review.
First submitted:
Accepted:
Reviewer comments
PDF: Download
DOI: 10.15347/WJM/2023.004
QID: Q115703404
XML: Download
Share article
![]() Email
|
Email
| ![]() Facebook
|
Facebook
| ![]() Twitter
|
Twitter
| ![]() LinkedIn
|
LinkedIn
| ![]() Mendeley
|
Mendeley
| ![]() ResearchGate
ResearchGate
Suggested citation format:
Vitalii Moskalov; Olena Koshova; Sabina Ali; Nataliia Filimonova (28 July 2023). "Impact of xenogenic mesenchymal stem cells secretome on a humoral component of the immune system". WikiJournal of Medicine 10 (1): 9. doi:10.15347/WJM/2023.004. Wikidata Q115703404. ISSN 2002-4436. https://upload.wikimedia.org/wikiversity/en/b/be/Impact_of_xenogenic_mesenchymal_stem_cells_secretome_on_a_humoral_component_of_the_immune_system.pdf.
License: ![]()
![]() This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
Athikhun Suwannakhan ![]() contact
contact
Article information
Abstract
Methods. The following methods were used in the study: isolation of mesenchymal stem cells from the bone marrow of cattle; culturing of isolated cells and obtaining a conditioned medium containing exometabolites with subsequent purification; modeling of secondary immunodeficiency in mice; setting up a test on mice to determine the number of antibody-forming cells in the spleen and hemagglutinin titers in blood serum; statistical data analysis.
Results. A study of the impact of the secretome of xenogenic mesenchymal stem cells (MSCs) on the humoral arm of immunity in mice with a normal immune status showed a significant increase in the antibody-forming cells count compared with the control by 3.4-8.8 times when administered intramuscularly and by 4.2-5.4 times when administered subcutaneously (at the same time, in the group of the reference drug, the increase was four times), as well as an increase in the titer of hemagglutinins concerning the control by 25-95% when administered intramuscularly and 32-52% when administered subcutaneously (the reference drug Thymalin was at the control level). With intramuscular administration, a clear relationship was observed between the concentration of MSC secretome and the biological effect (high significant positive correlation: ρ = 0.99, p ≤ 0.05 for calculating AFC; ρ = 0.97, p ≤ 0.05 for HA titer). Lethality in the group of animals what administered only hydrocortisone acetate (HCA, positive control) was 100%. The use of the mesenchymal stem cells secretome increased the survival of animals by 50% by stimulating the formation of the required number of antibody-forming cells and antibody titer, except for the subcutaneous route of administration (at the level of immunized control, animals with a normal immune status). The reference drug showed a result at a level significantly lower than the immunized control. The antibody titer with the subcutaneous route of administration of secretion of MSCs was significantly lower than the immunized control but significantly higher concerning the reference drug.
Conclusion. The administration of the secretome of xenogenic mesenchymal stem cells stimulated the humoral arm of immunity as same in mice with normal immune status as in mice with secondary immunodeficiency. The data obtained supplement the information on the introduction of live mesenchymal stem cells. Live allogeneic MSCs have a suppressive effect on B cells, while xenogenic MSCs cause a response on themselves. The secretome of xenogenic MSCs does not contain surface immunogenic molecules that are carried by living cells, but nevertheless increases the activity of the humoral component of immunity. The mechanisms of this effect require further study.
Introduction
Humoral immunity is a component of the immune system that is mediated by macromolecules of protein and peptide nature. The study of the dynamics and intensity of the immune macromolecules’ formation under the influence of various internal and external factors is one of the key problems of immunobiology. To assess the intensity of the formation of the humoral immune response, the antibody-forming cells (AFC, plasma cell) count and the antibody titer to the antigen with which the immunization was previously carried out can be determined.
Individual substances such as cytokines, in particular tumor necrosis factor (it enhances humoral responses to both T-independent and T-cell-dependent antigens by reducing apoptosis by binding to the CD40 ligand),[1] interleukin 12 (increases the production of almost all immunoglobulin isotypes),[2] complement system proteins (they are involved in labeling "foreign" pathogens and "presenting" them to B-lymphocytes, which enhances both antibody production and long-term memory),[3] some vitamins, in particular, vitamin E (stimulates the production of immunoglobulins G)[4] or mixtures of biologically active substances, for example, Echinacea purpurea (L.) Moench extract[5] can be used as stimulators of humoral immunity.
Mesenchymal stem cells are defined as clonogenic, non-hematopoietic stem cells present in adult tissues and capable of differentiating into mesodermal (e.g., osteoblasts, chondrocytes, endothelial cells) and non-mesoderm (neurons) cell lines. The relative ease of their isolation and their wide potential for differentiation, hypoimmunogenicity, and low ability to malignancy have long attracted the attention of researchers.[6] Research on the immunomodulatory properties of mesenchymal stem cells has mainly focused on their effects on T cells, although it is hypothesized that they may also affect B cells.[7] At the same time, the use of living cells has some limitations both in terms of their long-term storage (impossible without liquid nitrogen; freezing and thawing procedures are relatively difficult for consumers) and in terms of the risks of their use (although the risk of an immune response, malignancy, and abnormal differentiation is low, it`s different from zero). Such limitations are pushing researchers to actively study the biologically active substances produced by these cells, collectively referred to as the secretome. The secretome of mesenchymal stem cells has been described as a complex mixture of a soluble protein-peptide fraction (e.g., growth factors and cytokines) and a vesicular fraction containing proteins and genetic material (e.g., microRNAs).[8] It has significant therapeutic potential in connection, in particular, with the ability to "homing". Therefore, it is interesting to study the impact of the secretome of mesenchymal stem cells on the humoral arm of immunity.
Modulation of the immune system by mesenchymal stem cells is one of the key factors that determine their regenerative effect. In particular, it was established the role of MSCs in switching between M1 and M2 populations of macrophages; it causes the transition from an inflammatory response to clearing of cellular debris and healing.[9] MSCs can also influence the work of adaptive immunity, i.e. on activation, proliferation and differentiation of T- and B-lymphocytes. For example, dose-dependent inhibition of alloantigen-dependent T cell proliferation has been shown. Also, MSCs can stimulate regulatory T-type 1 (Tr1)-like cells, which are characterized by the secretion of interferon-γ (IFN-γ) and IL-10, that leads to an increase in populations of CD4+, CD25+, Foxp3+ and Tr1 cells and a decrease graft-versus-host disease. Also, data are described in the literature that MSCs significantly reduce the activity of antibody production by plasmacytes (derivatives of B cells) due to the suppression of chemokine receptors C-X-C type 4 (CXCR4), CXCR5 and CXCR7, as well as activation of transcription activator 3 (STAT3), and PAX5 expression.[10][11] MSCs also directly inhibit B cell activity via the PD-1/PD-L1 pathway. MSCs also promote the formation of non-activated B cells (naive, transitional, and memory subpopulations), which promote Treg differentiation. There was evidence that MSCs can induce the proliferation and differentiation of purified B cells. More recent studies have shown that suppression of B cells by MSCs requires signals from T cells.[11]
The objective of the study was to determine such parameters of humoral immunity as the number of antibody-forming cells and the titer of antibodies in the blood under the action of the whole fraction of the secretome of xenogenic mesenchymal stem cells in mice with a normal immune status and mice with secondary immunodeficiency caused by the hydrocortisone acetate.
Material and methods
Mesenchymal stem cells were obtained from the bovine bone marrow according to the standard procedure.[12] Bovine bone marrow was harvested from a healthy animal at the slaughterhouse and placed in Gibco™ phosphate-buffered saline. Cells were isolated from tissue samples under aseptic conditions in a laminar flow cabinet by washing with Gibco™ DMEM high glucose and stepwise purified using an MPW-56/10056 centrifuge (each centrifugation was carried out at 10 minutes 1000 rpm). The disaggregated cell suspension was plated on culture flask TPP in a medium containing 90% Gibco™ DMEM, high glucose, and 10% Biowest™ fetal bovine serum (country of origin – Colombia, supplier – BioloT™) and incubated at 37 ± 1 ºC and with 5.0 ± 0.2% CO2 in the atmosphere. After 24 hours of growth, the floating fraction was removed from the culture. Every 3-4 days, the medium was replaced with a fresh one. In the second passage the cells were cryopreserved according to the standard procedure.[13] The cell suspension was removed from the substrate using Gibco™ Trypsin/EDTA solution, purified using an MPW-56/10056 centrifuge (centrifugation was carried out at 10 minutes 1000 rpm), and added to a cryoprotective medium (90% Biowest™ fetal bovine blood serum, 10% Thermo Scientific™ DMSO), distributed between cryotubes (SPL Lifesciences) and cooled stepwise at a rate of 1ºC/min. The culture was restored after freezing in DMEM, high glucose Gibco™ nutrient medium supplemented with 10% Biowest™ fetal bovine blood serum and cultivated as before. After the second passage, the growth medium was replaced with a maintenance medium (with a content of serum reduced to 2 %). At 48 hours of culture growth, a conditioned medium containing the secretome of mesenchymal stem cells was collected (cell density was 5.5-6.0 million cells per 1.0 ml). The obtaining whole fraction of the secretome was purified from cell detritus using an MPW-56/10056 centrifuge (centrifugation was carried out at 10 minutes 1000 rpm) and used in further research. Thus, the secretome was obtained from mesenchymal stem cells of the second passage deconserved after low-temperature storage in liquid nitrogen, which grew for 48 hours on a maintenance medium containing 2% fetal bovine serum, in the form of a conditioned medium, purified from cell debris by centrifugation for 10 minutes at 1000 rpm.
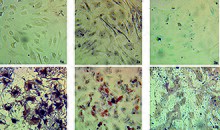
The belonging of the obtaining cell population to mesenchymal stem cells was confirmed according to three criteria: (1) adhesion to cultural plastic; (2) determination of the immunophenotype by the following markers on the cell surface: CD44 (97.9 ± 0.6)%, CD90 (99.3 ± 0.2)%, CD34 (3.4 ± 0.8)%; CD45 (2.7 ± 1.1)%; (3) three lineage differentiation after addition of a differentiation inducer for 21 days (osteogenic, adipogenic, and chondrogenic). Such differentiation inducers have been used: 10 mmol/l glycerol-2-phosphate, 50 μg/ml L-ascorbic acid and 100 nmol/l dexamethasone for osteogenesis; 0.2 mmol/l indomethacin, 0.5 mmol/l 3-isobutyl-1-methylxanthine, 0.01 mg/ml insulin and 1 µmol/l dexamethasone for adipogenesis; 0.1 µmol/l dexamethasone, 0.05 mmol/l ascorbic acid, 10 ng/ml TGF for chondrogenesis. The cells were incubated under standard conditions (temperature 37 ± 1 °C, with 5.0 ± 0.2% CO2 in the atmosphere). Every 3–4 days, the nutrient medium was changed to a fresh one without changing its composition. On the 21st day of incubation of mesenchymal stem cells with inducers, differentiation results were identified by staining.
The study was performed on white outbred adult mice of both sexes weighing 20 ± 2 g supplied by FOP Nechiporenko from supplier's own population of mice and used after quarantine for 14 days. All manipulations with animals complied with the requirements of international legislation for the protection of vertebrate animals[14] and the study was approved by the Bioethics Commission. The randomization was done by urn design taking into account the body weight of the animals (the deviation in the initial weight of animals did not exceed 10%). The animals were kept in a separate room at a temperature of 20-24°C, humidity 45-65%, the light regime "12 hours day/night", in individually ventilated plastic cages "Tecniplast", 6-10 animals each. The study was performed on mice with normal immune status (n=6) and with secondary immunodeficiency (n=10 at the beginning of the experiment). An immunodeficiency state (secondary immunodeficiency) in animals was induced by a single intraperitoneal injection of hydrocortisone acetate (HCA) at a dose of 250 mg/kg.[15] The whole fraction of the secretome of mesenchymal stem cells was studied with subcutaneous and intramuscular administration at concentrations of 10, 20, and 50 μl/kg (in terms of protein, 35, 70, and 175 μg/kg, respectively). The protein concentration in the secretome was determined for the accuracy of dosing samples by the spectrophotometric method by the difference in absorption intensity at wave lengths of 215 and 225 nm against saline under standard measurement conditions. As a reference drug, Thymalin (ZAT "Biofarma", Kyiv, Ukraine) was administered intramuscularly at a dose of 5 mg/kg. To determine the parameters of the humoral immune response, the animals were immunized with a 3% suspension of freshly washed ram erythrocytes. Accounting was carried out on the 5th day of immunization, which was carried out daily 1 hour before the administration of the test samples. In the control group, the animals were immunized with ram erythrocytes and injected with physiological saline intramuscularly. The impact of samples on the humoral arm of immunity (antibody formation processes) was assessed by the number of antibody-forming cells (AFC, plasma cell) in the spleen and hemagglutinin (HA) titers in the blood serum of mice. The amount of AFC in the spleen was determined by the method of of Jerne K. N. et al.[16] The method is based on the ability of lymphoid cells of animals immunized with alien erythrocytes to secrete anti-erythrocyte antibodies that cause antigen lysis in the presence of complement. According to the number of macroscopically visible zones of hemolysis around the AFC, the number of antibody producers per lymphoid organ was calculated. The HA titer in the blood serum of mice was studied in the agglutination reaction by gluing ram erythrocytes with antibodies in isotonic sodium chloride solution.[17] In an experiment with secondary immunodeficiency, samples were administered 5 days prior to the modeling of immunodeficiency and until the indicators were taken into account. Immunization with a 3% suspension of freshly washed ram erythrocytes was carried out for 4 days, starting from the next day after modeling the pathology. Accounting for survival was carried out before determining the amount of AFC and antibody titer on the next day after the completion of immunization (5th day after the administration of HCA) by taking into account the number of animals that had died by this time.
Statistical analysis performed using multiple tests by dint of Excel 2016. The Fisher's exact test for sample shares was applied to assess the differences between data expressed in %. Each sample of the dispersion complex was tested for normality of distribution using the Shapiro-Wilk W-test. If at least one of the samples of the dispersion complex had a distribution pattern different from normal (it was observed in all experiments), non-parametric tests were used: the Kruskal-Wallis test to detect the presence of a statistically significant difference between groups; the Mann–Whitney–Wilcoxon U-test to identify specific pairs of groups, the differences between which are significant. The Spearman's rank correlation coefficient was applied for assessment of the relationship between the parameters (the significance of the coefficient was assessed using Student's t-test).[18][19] The power of the statistical tests was estimated by dint of G*Power 3.1.9.7.[20]
Results
Three concentrations of the whole fraction (of 10, 20, and 50 μl/kg; in terms of protein, 35, 70, and 175 μg/kg, respectively) of mesenchymal stem cells secretome at two routes of administration (intramuscular and subcutaneous) were studied in mice with normal immune status during the study stage (fig. 2). Statistical analysis performed by the Kruskal-Wallis test showed a significant difference between the groups (p ≤ 0.001). Estimation of the antibody-forming cells (AFC) count (fig. 2a) showed their statistically significant increase compared to the control in the group of reference drug Thymalin by 4 times, as well as in all groups with different secretome concentrations with both routes of administration (3.4–8.8 times with the intramuscular route, and 4.2–5.4 times with subcutaneous one). At the same time, in groups with the maximum concentration of secretome (50 μl/kg) with both routes of administration, a statistically significant excess of the indicator compared to the reference drug group was observed. It should also be noted that at a concentration of 50 µl, the antibody-forming cells count depended on the route of administration: there was statistically significant excess in the indicator in the group with the intramuscular one (p ≤ 0.01, fig. 2a).
As for the hemagglutinin (HA) titers (fig. 2b), statistical analysis performed by the Kruskal-Wallis test also showed a significant difference between the groups (p ≤ 0.001). The control group and reference drug (Thymalin) group were on the same level by this indicator (differences between groups are not significant, p > 0.05). Among the experimental groups, the following picture was observed: all three concentrations with the subcutaneous administration were statistically significantly higher than the control group, but differed slightly from each other; with the intramuscular administration, only groups with concentrations of 20 and 50 μl/kg were statistically significantly higher than the control, but they significantly exceeded the group with the minimum concentration with this route of administration (by 27–57%). Experimental groups with a concentration of 50 µl/kg with different routes of administration, as in the case of the plasma cells count, were statistically significantly different: the intramuscular route of administration also turned out to be more effective than the subcutaneous one (there were significant differences in this indicator between groups receiving 50 µl/kg of MSCs secretome in two different routes, p≤0.01, fig. 2b).
Correlation analysis confirmed the presence of a high statistically significant positive relationship between the number of antibody-forming cells and hemagglutinin titer (ρ = 0.95; p ≤ 0.05, fig. 2c). The dose dependence of the observed effects on the humoral arm of immunity in mice with a normal immune status for intramuscular injection was also confirmed by correlation analysis of relationships between the studied parameters and secretome concentration (ρ = 0.99, p ≤ 0.05 for AFC count; ρ = 0.97, p ≤ 0.05 for HA titer). The relationship between the antibody-forming cells counts or hemagglutinin (HA) titers and the concentration of the secretome of mesenchymal stem cells is not statistically significant in the case of subcutaneous administration (the power of the statistical coefficient (< 80 %) is insufficient for the final acceptance of the null hypothesis of equality of samples).

The model of secondary immunodeficiency is a rigorous test of the immunomodulatory properties of substances, which makes it possible to assess the survival rate of the test animals and the compensatory properties of the immune system (in the experiment, they were assessed by the amount of AFC and antibody titer). The minimum concentration of the test substance causing an effect in animals with a normal immune status (10 µl/kg) was studied, other concentrations have not been studied at the moment. Secondary immunodeficiency caused by intraperitoneal administration of 250 mg/kg hydrocortisone acetate (HCA) resulted in 100% mortality (fig. 3a) in the absence of immunomodulating agents (positive control). The use of Thymalin and secretome of mesenchymal stem cells at a concentration of 10 μl/kg statistically significantly increased the survival of animals with secondary immunodeficiency (differences between groups with different immunomodulating agents are insignificant, p > 0.05).

Statistical analysis performed by the Kruskal-Wallis test showed a significant difference between the groups (p ≤ 0.001). In the groups of animals that were injected with the secretome of mesenchymal stem cells, the antibody-forming cells count (fig. 3b) was at the level of the immunized control (animals were immunized with ram erythrocytes to account immunity indicators; the pathology was not modeled). At the same time, in the reference drug (Thymalin) group, this indicator was statistically significantly lower than in the control. As for the antibody titer (fig 3c), the picture was the same with plasma cells count with the intramuscular administration; with the subcutaneous route of administration, the antibody titer was statistically significantly lower than in the control, but at the same time it significantly exceeded the reference drug group.
Correlation analysis confirmed the presence of a high statistically significant positive relationship between the number of antibody-forming cells and hemagglutinin titer (ρ = 0.90; p ≤ 0.05, fig 3d).
Discussion
Humoral immunity is also referred to as antibody-mediated immunity, so the key parameter of its performance is production of antibodies. This parameter can be assessed, for example, by the concentration and ratio of immunoglobulins in the blood serum. In this study, to assess antibody production, we used the count of the number of antibody-forming cells in the spleen and the titer of hemagglutenins after immunization of mice with 3% suspension of freshly washed ram erythrocytes.
A number of researchers note the immunosuppressive properties of allogeneic living MSCs on the production of antibodies. Comoli et al.[21] studied the impact of human mesenchymal stem cells (MSCs) on alloantigen-stimulated immunoglobulin production in an in vitro model of peripheral blood mononuclear cells cultured in mixed lymphocyte culture. It was shown that the addition of MSCs in a ratio of 4:1 and 20:1 has a strong inhibitory effect on the secretion of IgG, IgA, and IgM. These data are consistent with data on B cell inhibition obtained in animal models.[22][23] Mesenchymal stem cells-mediated inhibition of human B cell proliferation and antibody production in vitro after polyclonal stimulation has also been shown.[24] Comoli et al.[21] suggest that the inhibition of B cell function is predominantly due to the effect of suppressing T cell help, rather than a direct effect on B cell, and it is expressed in inhibition of the production of IL-5 and, possibly, IL-4, as well as downregulation of chemokine receptors on B cells. The researchers also note that although secreted factors play an important role in suppressing the production of antibodies, however, the inhibitory effect is higher when MSCs are in direct contact with immune cells.
On the other hand, there is evidence that xenogenic MSCs can enhance humoral responses. So, Jun-Man Hong et al.[25] note an increase in IgG1 and IgG2a titers in the blood serum of Balb/c and B6 mice (IgG2c instead of IgG2a in B6 mice) after the administration of human mesenchymal stem cells (hMSCs), moreover, the detected antibodies are specific to hMSCs. Analysis of the ratio of immunoglobulin subclasses and the morphology of germinal center formation in the mouse spleen indicates the manifestation of T-cell-dependent humoral responses to hMSC surface antigens. T-cell-dependent humoral responses involve the uptake of antigens by dendritic cells (DCs), their presentation by DCs to T lymphocytes, which, in turn, activate B cells and stimulate their differentiation into antibody-producing plasma cells, however, MSCs are able to inhibit the differentiation of DCs from progenitor cells, which can block the entire chain of processes. hMSCs also inhibited B cell differentiation. Observed by Jun-Man Hong et al.[25] enhancement of the humoral response under the conditions described is paradoxical and unresponsive, because ex-vivo activation of hMSCs by IFN-γ to model inflammation did not lead to immunosuppression, potential cytokine incompatibility between humans and mice is also not confirmed. At the same time, it should be noted that the secretome used in the work does not contain surface cellular molecules in significant amounts, therefore, it is less likely to cause an immune response to itself. Franka Luk et al. studied the immunomodulatory properties of heat shock-inactivated MSCs and concluded, in particular, that they do not induce the formation of regulatory B cells, unlike living cells.[26]
We have not found any literature data on the effect of the secretome of xenogenic MSCs on antibody production. Our study has shown the activation of the humoral arm of immunity after intramuscular (more pronounced) and subcutaneous administration of the whole fraction of the secretome produced by them. A fold increase in both the antibody-forming cells count in the spleen and serum antibody titer was shown after secretome administration at concentrations of 10, 20, and 50 μl/kg (in terms of protein, 35, 70, and 175 μg/kg, respectively). With intramuscular injection, a high and statistically significant dose dependence of the biological effect was observed. Subcutaneous injection of secretome showed a blurred picture of the biological effect. We can assume that with subcutaneous administration of the studied composition, paracrine effects prevail over systemic ones, which does not always allow achieving a generalized impact.
The model of secondary immunodeficiency used in the study by administering a high dose of hydrocortisone acetate is based on the depletion of immunocompetent cells in the thymus, spleen, femoral marrow, mesenteric, inguinal and popliteal lymph nodes after its application. In the early stages after hormone administration, B-lymphocytes are more affected (a sharp decrease in their number, for example, is observed in the spleen) than T-lymphocytes, which shifts the ratio of lymphocytes towards T cells. Also, T cells repair more quickly than B cells.[27] Compensation for the immunodeficiency state of the humoral component of adaptive immunity can be provided by cytoprotection or by activating the proliferation and differentiation of quiescent B cells. It has previously been shown that MSCs can affect B cells through in vitro cell-to-cell contact, which results in increasing survival of quiescent B cells through contact-dependent mechanisms, as well as enhancing B cell differentiation independently of T cells. MSCs also inhibit caspase 3-mediated B-cell apoptosis by enhancing vascular endothelial growth factor (VEGF), as well as slow down cell proliferation by blocking the cell cycle of B-lymphocytes in the G0/G1 phase by activating the p38 mitogen-activated protein kinases (MAPK) pathway. It is likely that the cytoprotective effect mediated by VEGF activation and impact on the MAPK pathway can be supported by components of the MSCs secretome, while the effects of contact cell interaction cannot be reproduced.[28]
The immune suppression provided by live auto- and allogeneic MSCs seems to be related to the characteristics of the niche in which they operate and their activation by inflammatory factors. The biological niche affects the activity of mesenchymal stem cells, the range of substances produced by them, and the set of surface molecules (for contact interaction). Upon obtaining the basic secretome in vitro conditions, in the absence of body factors influencing on the cells, its components stimulate antibody production. The described basic secretome also has a cytoprotective effect on spleen B cells, which was demonstrated in an experiment with secondary immunodeficiency. The mechanisms of the described facts require further study.
Conclusion
Administration of the whole fraction of MSCs secretome to mice with normal immune status resulted in a statistically significant increase in the antibody-forming cells count in the spleen and an increase in serum antibody titer. With the intramuscular injection, a high and statistically significant dose dependence of the biological effect was observed. In an experiment with a model of secondary immunodeficiency, the administration of the MSCs secretome increased the survival of animals and provided indicators of the humoral arm of immunity at the level of mice without immunodeficiency (except for subcutaneous administration). We assume that paracrine effects predominate over systemic ones with subcutaneous administration of the studied composition, so the effects may be lower. The data obtained supplement the information on the introduction of live mesenchymal stem cells. Live allogeneic MSCs have a suppressive effect on B cells, while xenogenic MSCs cause a response on themselves. The secretome of xenogenic MSCs does not contain surface immunogenic molecules that are carried by living cells, but nevertheless increases the activity of the humoral component of immunity. The mechanisms of this effect require further study.
Additional information
Acknowledgements
We appreciate researcher Alexey Lavrik for suggesting the idea for the study.
Competing interests
All authors declare that there is no conflict of interest.
Authors' contribution
Olena Koshova performed and supervised all tests on laboratory animals, the statistical analysis. Sabina Ali provided the isolation and cultivation of mesenchymal stem cells, the collection and purification of components of the secretome. Vitalii Moskalov has written most of the manuscript. Olena Koshova and Vitalii Moskalov has contributed assessing data and data collection and interpretation. Nataliia Filimonova and Irina Tishchenko selected the optimal research methods and participated in the discussion of the results. All authors participated in the design of the study and approved the final version of the manuscript.
Ethics statement
This study was approved by the Bioethics Commission of the Ministry of Health of Ukraine, National University of Pharmacy, Kharkiv, Ukraine (Minutes No. 6 dated 06/20/2018)
References
- ↑ Do, Richard K.G.; Hatada, Eunice; Lee, Hayyoung; Tourigny, Michelle R.; Hilbert, David; Chen-Kiang, Selina (2000-10-02). "Attenuation of Apoptosis Underlies B Lymphocyte Stimulator Enhancement of Humoral Immune Response". Journal of Experimental Medicine 192 (7): 953–964. doi:10.1084/jem.192.7.953. ISSN 0022-1007. PMID 11015437. PMC PMC2193312. https://rupress.org/jem/article/192/7/953/8239/Attenuation-of-Apoptosis-Underlies-B-Lymphocyte.
- ↑ Metzger, Dennis W.; Buchanan, J. Matthew; Collins, John T.; Lester, Terry L.; Murray, Kristin S.; Cleave, Victor H.; Vogel, Laura A.; Dunnick, Wesley A. (1996-10). "Enhancement of Humoral Immunity by Interleukin-12". Annals of the New York Academy of Sciences 795 (1): 100–115. doi:10.1111/j.1749-6632.1996.tb52659.x. ISSN 0077-8923. https://onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.1996.tb52659.x.
- ↑ Carroll, Michael C. (2008-12). "Complement and humoral immunity". Vaccine 26: I28–I33. doi:10.1016/j.vaccine.2008.11.022. PMID 19388161. PMC PMC4018718. https://linkinghub.elsevier.com/retrieve/pii/S0264410X08015235.
- ↑ Tengerdy, R.P.; Heinzerling, R.H.; Brown, G.L.; Mathias, M.M. (1973). "Enhancement of the Humoral Immune Response by Vitamin E". International Archives of Allergy and Immunology 44 (2): 221–232. doi:10.1159/000230931. ISSN 1018-2438. https://www.karger.com/Article/FullText/230931.
- ↑ Freier, David O.; Wright, Kevin; Klein, Kyra; Voll, Darrell; Dabiri, Kofoworola; Cosulich, Katherine; George, Ryan (2003-01). "Enhancement of the Humoral Immune Response by Echinacea purpurea in Female Swiss Mice". Immunopharmacology and Immunotoxicology 25 (4): 551–560. doi:10.1081/IPH-120026440. ISSN 0892-3973. http://www.tandfonline.com/doi/full/10.1081/IPH-120026440.
- ↑ Kassem, Moustapha; Kristiansen, Malthe; Abdallah, Basem M. (2004-11). "Mesenchymal Stem Cells: Cell Biology and Potential Use in Therapy". Basic Clinical Pharmacology Toxicology 95 (5): 209–214. doi:10.1111/j.1742-7843.2004.pto950502.x. ISSN 1742-7835. https://onlinelibrary.wiley.com/doi/10.1111/j.1742-7843.2004.pto950502.x.
- ↑ Franquesa, Marcella; Hoogduijn, M. J.; Bestard, O.; Grinyó, J. M. (2012). "Immunomodulatory Effect of Mesenchymal Stem Cells on B Cells". Frontiers in Immunology 3. doi:10.3389/fimmu.2012.00212. ISSN 1664-3224. PMID 22833744. PMC PMC3400888. http://journal.frontiersin.org/article/10.3389/fimmu.2012.00212/abstract.
- ↑ Teixeira, Fábio G.; Salgado, António J. (2020). "Mesenchymal stem cells secretome: current trends and future challenges". Neural Regeneration Research 15 (1): 75. doi:10.4103/1673-5374.264455. ISSN 1673-5374. PMID 31535654. PMC PMC6862404. https://journals.lww.com/10.4103/1673-5374.264455.
- ↑ Aurora, Arin B.; Olson, Eric N. (2014-07). "Immune Modulation of Stem Cells and Regeneration". Cell Stem Cell 15 (1): 14–25. doi:10.1016/j.stem.2014.06.009. PMID 24996166. PMC PMC4131296. https://linkinghub.elsevier.com/retrieve/pii/S1934590914002574.
- ↑ González-González, Alberto; García-Sánchez, Daniel; Dotta, Monica; Rodríguez-Rey, José C; Pérez-Campo, Flor M (2020-12-26). "Mesenchymal stem cells secretome: The cornerstone of cell-free regenerative medicine". World Journal of Stem Cells 12 (12): 1529–1552. doi:10.4252/wjsc.v12.i12.1529. ISSN 1948-0210. PMID 33505599. PMC PMC7789121. https://www.wjgnet.com/1948-0210/full/v12/i12/1529.htm.
- ↑ 11.0 11.1 Jiang, Wei; Xu, Jianyong (2020-01). "Immune modulation by mesenchymal stem cells". Cell Proliferation 53 (1). doi:10.1111/cpr.12712. ISSN 0960-7722. PMID 31730279. PMC PMC6985662. https://onlinelibrary.wiley.com/doi/10.1111/cpr.12712.
- ↑ Colter, David C.; Class, Reiner; DiGirolamo, Carla M.; Prockop, Darwin J. (2000-03-28). "Rapid expansion of recycling stem cells in cultures of plastic-adherent cells from human bone marrow". Proceedings of the National Academy of Sciences 97 (7): 3213–3218. doi:10.1073/pnas.97.7.3213. ISSN 0027-8424. https://pnas.org/doi/full/10.1073/pnas.97.7.3213.
- ↑ Freshney, R. Ian (2016). Culture of animal cells: a manual of basic technique and specialized applications (7th ed.). Hoboken, New Jersey: Wiley-Blackwell. ISBN 978-1-118-87337-3. OCLC 956711701. https://www.worldcat.org/oclc/956711701.
- ↑ "EUR-Lex - 31986L0609 - EN - EUR-Lex". eur-lex.europa.eu. Retrieved 2022-10-04.
- ↑ van Ogtrop, M L; Mattie, H; Sekh, B R; van Strijen, E; van Furth, R (1992-11). "Comparison of the antibacterial efficacies of ampicillin and ciprofloxacin against experimental infections with Listeria monocytogenes in hydrocortisone-treated mice". Antimicrobial Agents and Chemotherapy 36 (11): 2375–2380. doi:10.1128/AAC.36.11.2375. ISSN 0066-4804. PMID 1489180. PMC PMC284338. https://journals.asm.org/doi/10.1128/AAC.36.11.2375.
- ↑ Jerne, Niels K.; Nordin, Albert A. (1963-04-26). "Plaque Formation in Agar by Single Antibody-Producing Cells". Science 140 (3565): 405–405. doi:10.1126/science.140.3565.405.a. ISSN 0036-8075. https://www.science.org/doi/10.1126/science.140.3565.405.a.
- ↑ Feizi, T.; Monger, E. (1970-01-02). "Microtitration of Serum Cold Agglutinins". Transfusion 10 (1): 33–35. doi:10.1111/j.1537-2995.1970.tb00700.x. https://onlinelibrary.wiley.com/doi/10.1111/j.1537-2995.1970.tb00700.x.
- ↑ Atramentova, L. A.; Utevskaia, O. M. (2008). Statisticheskie metody v biologii uchebnik [Statistical methods in biology: textbook] (in Russian). Gorlovka: Vydavnytstvo Likhtar. ISBN 978-966-2129-26-7.
- ↑ Lapach, S. N.; Chubenko, A. V.; Babich, P. N. (2001). Statisticheskie metody v mediko-biologicheskikh issledovaniiakh s ispolzovaniem Excel [Statistical methods in biomedical research using Excel] (in Russian) (2nd ed.). Kiev: Morion. ISBN 966-7632-33-4.
- ↑ Mayr, Susanne; Erdfelder, Edgar; Buchner, Axel; Faul, Franz (2007-09-01). "A short tutorial of GPower". Tutorials in Quantitative Methods for Psychology 3 (2): 51–59. doi:10.20982/tqmp.03.2.p051. ISSN 1913-4126. http://www.tqmp.org/RegularArticles/vol03-2/p051.
- ↑ 21.0 21.1 Comoli, P.; Ginevri, F.; Maccario, R.; Avanzini, M. A.; Marconi, M.; Groff, A.; Cometa, A.; Cioni, M. et al. (2007-01-08). "Human mesenchymal stem cells inhibit antibody production induced in vitro by allostimulation". Nephrology Dialysis Transplantation 23 (4): 1196–1202. doi:10.1093/ndt/gfm740. ISSN 0931-0509. https://academic.oup.com/ndt/article-lookup/doi/10.1093/ndt/gfm740.
- ↑ Glennie, Sarah; Soeiro, Inês; Dyson, Peter J.; Lam, Eric W.-F.; Dazzi, Francesco (2005-04-01). "Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells". Blood 105 (7): 2821–2827. doi:10.1182/blood-2004-09-3696. ISSN 0006-4971. https://ashpublications.org/blood/article/105/7/2821/20209/Bone-marrow-mesenchymal-stem-cells-induce-division.
- ↑ Augello, Andrea; Tasso, Roberta; Negrini, Simone Maria; Amateis, Andrea; Indiveri, Francesco; Cancedda, Ranieri; Pennesi, Giuseppina (2005-05). "Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway". European Journal of Immunology 35 (5): 1482–1490. doi:10.1002/eji.200425405. ISSN 0014-2980. https://onlinelibrary.wiley.com/doi/10.1002/eji.200425405.
- ↑ Corcione, Anna; Benvenuto, Federica; Ferretti, Elisa; Giunti, Debora; Cappiello, Valentina; Cazzanti, Francesco; Risso, Marco; Gualandi, Francesca et al. (2006-01-01). "Human mesenchymal stem cells modulate B-cell functions". Blood 107 (1): 367–372. doi:10.1182/blood-2005-07-2657. ISSN 0006-4971. https://ashpublications.org/blood/article/107/1/367/21754/Human-mesenchymal-stem-cells-modulate-Bcell.
- ↑ 25.0 25.1 Hong, Jun-Man; Kim, Jin-Hee; Kim, Gwang-Hoon; Shin, Hyun-Mu; Hwang, Young-il (2022-08-30). "Xenogeneic Humoral Immune Responses to Human Mesenchymal Stem Cells in Mice". International Journal of Stem Cells 15 (3): 291–300. doi:10.15283/ijsc21116. ISSN 2005-5447. PMID 34965998. PMC PMC9396016. http://www.ijstemcell.com/journal/view.html?doi=10.15283/ijsc21116.
- ↑ Luk, Franka; de Witte, Samantha F.H.; Korevaar, Sander S.; Roemeling-van Rhijn, Marieke; Franquesa, Marcella; Strini, Tanja; van den Engel, Sandra; Gargesha, Madhusudhana et al. (2016-09-15). "Inactivated Mesenchymal Stem Cells Maintain Immunomodulatory Capacity". Stem Cells and Development 25 (18): 1342–1354. doi:10.1089/scd.2016.0068. ISSN 1547-3287. https://www.liebertpub.com/doi/10.1089/scd.2016.0068.
- ↑ Dracott, B. N.; Smith, C. E. (1979-10). "Hydrocortisone and the antibody response in mice. I. Correlations between serum cortisol levels and cell numbers in thymus, spleen, marrow and lymph nodes". Immunology 38 (2): 429–435. ISSN 0019-2805. PMID 511224. PMC 1457917. https://pubmed.ncbi.nlm.nih.gov/511224.
- ↑ Song, Na; Scholtemeijer, Martijn; Shah, Khalid (2020-09). "Mesenchymal Stem Cell Immunomodulation: Mechanisms and Therapeutic Potential". Trends in Pharmacological Sciences 41 (9): 653–664. doi:10.1016/j.tips.2020.06.009. PMID 32709406. PMC PMC7751844. https://linkinghub.elsevier.com/retrieve/pii/S0165614720301450.
