WikiJournal of Medicine/History of penicillin
WikiJournal of Medicine
Open access • Publication charge free • Public peer review • Wikipedia-integrated
This article has been through public peer review.
It was adapted from the Wikipedia page History_of_penicillin and contains some or all of that page's content licensed under a CC BY-SA license. Post-publication review comments or direct edits can be left at the version as it appears on Wikipedia.
First submitted:
Accepted:
Reviewer comments
PDF: Download
DOI: 10.15347/WJM/2021.003
QID: Q107303937
XML: Download
Share article
![]() Email
|
Email
| ![]() Facebook
|
Facebook
| ![]() Twitter
|
Twitter
| ![]() LinkedIn
|
LinkedIn
| ![]() Mendeley
|
Mendeley
| ![]() ResearchGate
ResearchGate
Suggested citation format:
Kholhring Lalchhandama (22 October 2021). "History of penicillin". WikiJournal of Medicine 8 (1): 3. doi:10.15347/WJM/2021.003. Wikidata Q107303937. ISSN 2002-4436. https://upload.wikimedia.org/wikiversity/en/1/1c/History_of_Penicillin.pdf.
Citation metrics
AltMetrics
Page views on Wikipedia
Wikipedia: This work is adapted from the Wikipedia article History of penicillin (CC BY-SA). Content has also subsequently been used to update that same Wikipedia article History of penicillin.
License: CC-BY-SA
Editors:Roger Watson ![]() (handling editor) contact
(handling editor) contact
Mitchell Hammond
Luz Maria Hernandez-Saenz
Article information
Abstract
Early history

Yikrazuul, Public domain
Penicillin (Figure 1) is the second antibiotic and the first naturally-occurring antibiotic discovered.[1][2] The first antibiotic discovered was arsphenamine, marketed as Salvarsan, by German physician Paul Ehrlich and his Japanese assistant Sahachiro Hata in 1909.[3] It was a modified compound of a highly toxic chemical arsenic[4] that was used for the treatment of sexually transmitted bacterial (Treponema pallidum) infection or syphilis, and became the most commonly prescribed drug in the early 20th century.[5] However, it was overshadowed by penicillin, a safer and more efficacious antibiotic, that was effective against a wide range of Gram-positive bacteria,[6] as well as Gram-negative T. pallidum.[7]
Traditional curative practices preceded the discovery of penicillin as a component of the mould Penicillium (from the Latin word penicillum, meaning "painter's brush").[8] Ancient Egypt, Greece and India were aware of the curative properties of fungi and plants in treating bacterial infections,[9] as shown by the 16th-century BCE record of a Greek king of the use of bread moulds by a woman healer to treat wounded soldiers. Around the same time, Chinese traditional practitioners used moulds from soya bean for wound infections.[10]
In 17th-century Poland, wet bread was mixed with spider webs (which often contained fungal spores) to treat wounds, a technique mentioned by Henryk Sienkiewicz in his 1884 book With Fire and Sword. In 1640, the idea of using mould as a form of medical treatment was recorded by English apothecaries such as John Parkinson, royal botanist to Charles I, who described the use of certain mould (possibly Penicillium) in his book on pharmacology Theatrum Botanicum (The Botanical Theatre).[11][12] One of the common practices for treating impetigo (an infection due to the bacterium Staphylococcus aureus) was mould therapy using moulds obtained from bread and porridge.[13] A Canadian biologist A. E. Cliffe provided a vivid description:
It was during a visit through central Europe in 1908 that I came across the fact that almost every farmhouse followed the practice of keeping a mouldy loaf on one of the beams in the kitchen. When I asked the reason for this I was told that this was an old custom and that when any member of the family received an injury such as a cut or bruise, a thin slice from the outside of the loaf was cut off, mixed into a paste with water and applied to the wound with a bandage. It was assumed that no infection would result from such a cut.[14]
One of the most detailed medical narratives was how Brenda Ward (née Whitnear) was cured of her facial impetigo in 1929. After treating an eight-year-old Brenda with all possible medications available, the family physician James Twomey resorted to traditional practice and advised the mother to prepare a starch paste. The paste was was left in the pantry kept at the cellar head for several days until it became very mouldy. It was then applied on the girl's face as an ointment for over a week until she was completely healed.[15] There is no written record of the treatment except for the receipt of the consultation fee. Ward recalled that the mould initially appeared yellow in colour, grew into bronze colour, and finally turned into blue-green colonies, which indicates it was either Penicillium or Aspergillus. Based on Ward's description, in 1989, British microbiologist Milton Wainright concluded that most likely the mould was Penicillum due the growth pattern and antibacterial activity.[16]
Traditional treatments often worked because numerous organisms, including many species of moulds, naturally produce antibiotic substances. However, it was not until recently that practitioners were able to identify or isolate the active components in these organisms.[17]
Early scientific evidence
The modern history of penicillin research began in earnest in the 1870s in the United Kingdom. Sir John Scott Burdon-Sanderson, physiologist and lecturer at St. Mary's Hospital, observed that culture fluid covered with mould inhibited bacterial growth in his experiments on spontaneous generation in 1870.[18] His reports in 1871 described:
On October 10 glass a was turbid, and was found on microscopical examination to be teeming with bacteria; a thick whitish scum had formed on its surface. Glass h was perfectly clear; there were, however, great numbers of torula [a type of yeast] cells on its surface, but no bacteria. On October 12 6 exhibited numerous tufts of penicillium, but the liquid still remained limpid and free from bacteria.[19][a]
From his experiments, Burdon-Sanderson believed that decomposition or putrefaction was caused by bacteria, and not by moulds. He applied the Penicillium mould on dissected thigh muscles from a guinea pig and left them in bell jars. After two weeks tissues in sterilised jars and with Penicillium mould did not show signs of rotting. These experiments indicate that the mould had antibacterial activity, but Burdon-Sanderson failed to notice the importance of this finding.[20]
Burdon-Sanderson's discovery prompted Joseph Lister, an English surgeon and the father of modern antisepsis, to discover in 1871 that urine samples contaminated with mould also prevented the growth of bacteria.[21] Lister identified the mould as Penicillium glaucum and found that most bacteria could not grow alongside this mould.[22] He also described the antibacterial action of the mould on human tissue.[23] In 1877, he treated Ellen Jones, a nurse at King's College Hospital, whose wounds did not respond to any traditional antiseptic. The nurse was cured when a crude extract of P. glaucum culture was applied to her wounds. Uncertain of the nature and effect of the mould, Lister did not published his observations.[20]
In 1873, Welsh physician William Roberts, who later coined the term "enzyme", also conducted experiments on spontaneous generation and observed that glass tubes were easily contaminated by airborne bacteria and moulds.[24] In his 1874 report in the Philosophical Transactions of the Royal Society, he stated: "I have repeatedly observed that liquids in which the Penicillum glaucum was growing luxuriantly could with difficulty be artificially infected with Bacteria; it seemed, in fact, as if this fungus played the part of the plants in an aquarium, and held in check the growth of Bacteria, with their attendant putrefactive changes."[25] This is regarded as "the first unequivocal published statement" on antibacterial activity of any substance.[20]
John Tyndall, professor of physics at the Royal Institution of Great Britain, followed up on Roberts's work on refutation of spontaneous generation and demonstrated in 1875 the antibacterial action of the P. glaucum. His report, read before the Royal Society in 1876 (and published as a monograph in 1881),[26] stated:
[The] two most actively charged tubes were in part crowned by beautiful tufts of Penicilllum Glaucum. This expanded gradually until it covered the entire surface with a thick tough layer, which must have seriously intercepted the oxygen necessary to the Bacterial life. The bacteria lost their translatory power, fell to the bottom, and left the liquid between them and the superficial layer clear.[27]
In 1876, German biologist Robert Koch discovered that Bacillus anthracis was the causative pathogen of anthrax;[28] it was the first time a specific bacterium was proved to cause a specific disease, and the first direct evidence of the germ theory of diseases.[29] A year later, French biologists Louis Pasteur and Jules Francois Joubert observed that, when contaminated with moulds, cultures of the anthrax bacilli could be successfully inhibited.[30] They reported their findings in the Comptes Rendus de l'Académie des Sciences:
Neutral or slightly alkaline urine is an excellent medium for the bacteria... But if when the urine is inoculated with these bacteria an aerobic organism, for example one of the "common bacteria," is sown at the same time, the anthrax bacterium makes little or no growth and sooner or later dies out altogether. It is a remarkable thing that the same phenomenon is seen in the body even of those animals most susceptible to anthrax, leading to the astonishing result that anthrax bacteria can be introduced in profusion into an animal, which yet does not develop the disease; it is only necessary to add some "common 'bacteria" at the same time to the liquid containing the suspension of anthrax bacteria. These facts perhaps justify the highest hopes for therapeutics.[31]
The phenomenon was described by Pasteur and Koch as antibacterial activity and was named as "antibiosis" by French biologist Jean Paul Vuillemin in 1877[32][33] (the term antibiosis, meaning "against life", was adopted as "antibiotic" by American biologist and later Nobel laureate Selman Waksman in 1947[34]). It has also been asserted that Pasteur identified the mould as Penicillium notatum. However, Paul de Kruif's 1926 Microbe Hunters disagrees, describing this incident as contamination by other bacteria rather than by mould.[35] Ten years later, in 1887, Swiss physician Carl Alois Philipp Garré developed a test method using glass plate to see bacterial inhibition and found similar results.[33] Using a gelatin-based culture plate, he grew two different bacteria and found that their growths were inhibited differently; and reported:
I inoculated on the untouched cooled [gelatin] plate alternate parallel strokes of B. fluorescens [Pseudomonas fluorescens] and Staph. pyogenes [Streptococcus pyogenes ]... B. fluorescens grew more quickly... [This] is not a question of overgrowth or crowding out of one by another quicker-growing species, as in a garden where luxuriantly growing weeds kill the delicate plants. Nor is it due to the utilization of the available foodstuff by the more quickly growing organisms, rather there is an antagonism caused by the secretion of specific, easily diffusible substances which are inhibitory to the growth of some species but completely ineffective against others.[31]
At the University of Naples, in 1895, physician Vincenzo Tiberio published his research about moulds initially found in a water well in Arzano.[36] He noticed that whenever the moulds on the wall of the well was cleaned, many people had abdominal pain due to infection (enteritis), which was unheard of when the well was mouldy. He believed that there was a link between the infection and the mould.[37] After identifying different moulds from the well, he found that Penicilium and Aspergillus could kill certain bacteria including Vibrio cholerae, and some staphylococci strains.[38] From his observations, he concluded that these moulds contained soluble substances having antibacterial action.[38] However, his findings did not receive any attention until the discovery of penicillin.[37]
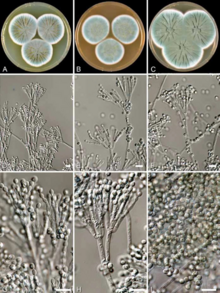
Houbraken et al., 2011, CC-BY 4.0
French medical student Ernest Duchesne at École du Service de Santé Militaire (Military Service Health School) in Lyon independently discovered the healing properties of P. glaucum.[39] He was able to grow the mould on pieces of moist food. When he mixed the mould with the bacterium Escherichia coli, he found that the bacteria did not grow, and when he injected the mould juice into guinea pigs experimentally inoculated with typhoid bacteria (Salmonella enterica), the animals never developed the disease.[40] He described the experiment in his 1897 doctoral dissertation titled Contribution à l'étude de la concurrence vitale chez les microorganismes (Contribution to the study of vital competition between microorganisms: antagonism between moulds and microbes) submitted to the Pasteur Institute.[41][42] Unfortunately, his discovery was ignored by the institute and soon forgotten. It was not until 50 years later when a librarian found the thesis, once penicillin had already been discovered.[43]
Duchesne could not continue his experiments due to a severe illness (believed to be tuberculosis) he contracted five years later. He died in 1912 while serving in the French Army.[40] He was himself using moulds to treat horses, a method learned from Arab stable boys to cure animals' sores; but he did not claim that the mould contained any antibacterial substance, only that it somehow protected the animals.[30] His conclusion was nonetheless prognostic, stating that competition between bacteria and moulds could be useful in the medical management of infections.[44] Penicillin does not cure typhoid as it is ineffective against most Gram-negative bacteria[45] and so it remains unknown which substance might have been responsible for Duchesne's cure.[b] A similar antibiotic effect of Penicillium was recorded in 1923 by Costa Rican Clodomiro Picado Twight, a Pasteur Institute scientist. In these early stages of penicillin research, most species of Penicillium were non-specifically referred to as P. glaucum, so that it is impossible to know the exact species and that it was really penicillin that prevented bacterial growth.[30]
The first to discover and isolate an antibiotic compound from Penicillium was an Italian physician Bartolomeo Gosio.[46] Gosio was investigating pellagra, which at the time was a common disease in southern Europe and America. It was known that the staple food of people having the disease was corn, and fungal contamination of corn was regarded as the source (American biochemist Conrad Elvehjem would identify in 1937 its aetiology as the deficiency of niacin or vitamin B3.[47]) In 1893, Gosio identified the mould Penicillium brevicompactum as one possible cause,[48] developed a simple culture method to make pure culture extract in crystalline form.[49] In 1896, he tested the substance on anthrax bacillus and found that it was highly potent against the bacteria.[50][51] Nonetheless, his discovery was largely forgotten as the substance was found not to be the cause of pellagra, and its medicinal potential was not obvious. American scientists, Carl Alsberg and Otis Fisher Black resynthesized Gosio's substance in 1912 giving it the name mycophenolic acid, which is now used as an immunosuppressant.[49][52]
In 1924, Andre Gratia and Sara Dath at the Free University of Brussels, Belgium, found that dead Staphylococcus aureus cultures were contaminated by a mould, a streptomycete. On further experimentation, they showed that the mould extract could kill not only S. aureus, but also Pseudomonas aeruginosa, Mycobacterium tuberculosis and Escherichia coli.[53] Gratia called the antibacterial agent "mycolysate" (killer mould). The next year they found another killer mould that could inhibit anthrax bacterium (B. anthracis). Reporting in Comptes Rendus Des Séances de La Société de Biologie et de Ses Filiales, they identified the mould as Penicillium glaucum.[54] In 1927, Gratia reported its medical use:
A poor patient who during three years had suffered from furuncles [infection by S. aureus], in spite of all treatments, was sent to us in despair. Jaumain did not hesitate to continue the treatment by a series of injections of the mycolysat. The result was remarkable. Not only was the recovery rapid, but it is now three years that [sic] this recovery continues without the slightest relapse. Since that time we have given the mycolysat to a very large number of cases. It is the most effective treatment even of the most resistant types of staphylococcic diseases.[46]
Unfortunately, as in the case of Duchesne, these findings received little attention as the antibacterial agent and its medical values were not fully understood; moreover, Gratia's samples were lost.[53]
The breakthrough discovery
Background

Ministry of Information Photo Division Photographer, Public domain
Penicillin as we know it today was "accidentally" discovered by the Scottish physician Alexander Fleming in 1928.[55][56] While working at St Mary's Hospital, London, Fleming was investigating the pattern of variation in S. aureus (Figures 2 and 3).[57] He was inspired by the recent discovery by the Irish physician Joseph Warwick Bigger and his two students C.R. Boland and R.A.Q. O’Meara at the Trinity College, Dublin, Ireland, in 1927. Bigger and his students found that a particular strain of S. aureus (designated "Y") that they isolated a year before from the pus of an axillary abscess (boil on the armpit) from one individual grew into a variety of strains. They published their discovery as "Variant colonies of Staphylococcus aureus" in The Journal of Pathology and Bacteriology, by concluding:
We were surprised and rather disturbed to find, on a number of plates, various types of colonies which differed completely from the typical aureus colony. Some of these were quite white; some, either white or of the usual colour were rough on the surface and with crenated margins.[58]
Fleming and his research scholar Daniel Merlin Pryce pursued this experiment but Pryce was transferred to another laboratory in early 1928. Their experiment had been successful and Fleming was planning and agreed to write a report in the compendium A System of Bacteriology to be published by the Medical Research Council at the end of that year.[57]
Initial discovery
In August, Fleming spent a vacation with his family at his country home The Dhoon at Barton Mills, Suffolk. Before leaving his laboratory (Figures 3 & 4), he inoculated several culture plates with S. aureus. He kept the plates aside on one corner of the table away from direct sunlight to allow proper growth of the bacteria. There are two versions of the reason he returned to his laboratory during the vacation. According to some sources, he was appointed Professor of Bacteriology at the St Mary's Hospital Medical School and had to officially join on Saturday 1 September 1928. The next Monday he visited the laboratory.[57][59] Alternative sources say that he went to London "on a flying visit" to help a colleague with the treatment of haemolytic bacillus. It was while waiting for the colleague that he visited the laboratory.[60][61] At the laboratory, Pryce paid him a visit and found him sorting out the Petri dishes. He and Pryce noticed one culture plate with an open lid and the culture contaminated with a blue-green mould. In the contaminated plate the bacteria around the mould did not grow, while those farther away grew normally, meaning that the mould killed the bacteria. Fleming commented as he watched the plate: "That's funny."[62] Pryce remarked to Fleming: "That's how you discovered lysozyme."[61]
Experiment

Vera de Kok, CC-BY 4.0
Fleming went off to resume his vacation and returned to his laboratory late in September.[57] He collected the original mould and grew it in culture plates. After four days he found that the plates developed large colonies of the mould. He repeated the experiment with the same bacteria-killing results. He later recounted his experience:
When I woke up just after dawn on September 28, 1928, I certainly didn't plan to revolutionize all medicine by discovering the world's first antibiotic, or bacteria killer. But I suppose that was exactly what I did.[63]
He concluded that the mould was releasing a substance that was inhibiting bacterial growth, and he produced a culture broth of the mould and subsequently concentrated the antibacterial component.[64] After testing against different bacteria, he found that the mould could kill only specific bacteria. For example, Gram-positive bacteria such as Staphylococcus, Streptococcus, and diphtheria bacillus (Corynebacterium diphtheriae) were easily killed; but there was no effect on Gram-negative species like typhoid bacterium (Salmonella typhimurium) and influenza bacillus (Haemophilus influenzae). He prepared a large-culture method from which he could obtain large amounts of the mould juice. On 7 March 1929, he coined the name "penicillin" for the mould extract,[59] explaining the reason as "to avoid the repetition of the rather cumbersome phrase 'Mould broth filtrate', the name 'penicillin' will be used."[65] In his Nobel lecture, he gave a further explanation:
I have been frequently asked why I invented the name "Penicillin". I simply followed perfectly orthodox lines and coined a word which explained that the substance penicillin was derived from a plant of the genus Penicillium just as many years ago the word "Digitalin" was invented for a substance derived from the plant Digitalis.[66]
Fleming had no training in chemistry so that he left all the chemical works to his new research scholar Stuart Craddock, who joined him in December;[60] he once remarked: "I am a bacteriologist, not a chemist."[57] In January 1929, Fleming recruited his former research scholar Frederick Ridley, who had studied biochemistry, to study the chemical properties of the mould.[62] However, both Craddock and Ridley left Fleming for other jobs before completing the experiments and isolating penicillin.[59] Their failure to isolate the compound resulted in Fleming practically abandoning further research on the chemical aspects of penicillin.[67] Nonetheless, he continued doing biological tests up to 1939.[59]
Identification of the mould

Houbraken et al., 2011, CC-BY 4.0
The source of the fungal contamination in Fleming's experiment remained a matter of speculation for several decades. The Royal Society of Chemistry believed that it came from a cup of coffee which Fleming left on the table.[43] In 1945, Fleming himself suggested that the fungal spores came through the window facing Praed Street. This story was regarded as a reliable explanation and was popularised in the literature,[60] starting with George Lacken's 1945 book The Story of Penicillin.[59] But it was later disputed by his co-workers, particularly Pryce, who testified much later that Fleming's laboratory window was kept shut all the time.[61] Ronald Hare also agreed in 1970 that the window remain locked as a large table in front of it made it difficult to open. In 1966, Charles John Patrick La Touche told Hare that he had given Fleming 13 specimens of fungi (10 from his lab) and only one from his lab was showing penicillin-like antibacterial activity.[60] It was therefore concluded that Fleming's mould came from La Touche's lab, located a floor below, and the spores had drifted in the air through the open doors.[68]
After a structural comparison with different species of Penicillium, Fleming believed that his specimen was Penicillium chrysogenum, a species described by the American microbiologist Charles Thom in 1910. He was fortunate as Charles John Patrick La Touche, an Irish botanist, had just recently joined as a mycologist at St Mary's to investigate fungi as the cause of asthma.[69] La Touche identified the specimen as Penicillium rubrum,[70] the identification used by Fleming in his publication of the discovery.[71]
In 1931, Thom re-examined different species of Penicillium including that of Fleming's specimen. He came to a confusing conclusion, stating: "Ad. 35 [Fleming's specimen] is P. notatum WESTLING. This is a member of the P. chrysogenum series with smaller conidia than P. chrysogenum itself."[72] From then on, Fleming's mould was synonymously referred to as P. chrysogenum and P. notatum, a species discovered by Swedish chemist Richard Westling in 1811. But Thom adopted and popularised the use of P. chrysogenum.[73] Adding to the controversial taxonomy, newly discovered species such as P. meleagrinum and P. cyaneofulvum were recognised as members of P. chrysogenum in 1977.[74] To resolve the confusion, in 2005, the Seventeenth International Botanical Congress held in Vienna, Austria, formally adopted the name P. chrysogenum as the conserved name (nomen conservandum) for all related Penicilium species.[75] In 2011, whole genome sequence and phylogenetic analysis revealed that Fleming's mould belongs to P. rubens (Figure 5), a species described by Belgian microbiologist Philibert Biourge in 1923, and that P. chrysogenum is a different species.[76][77]
Reception and publication
Initially, Fleming's discovery was not considered important, and as he showed to his his colleagues, all he received was an indifferent response. He described the discovery on 13 February 1929 before the Medical Research Club but his presentation titled "A medium for the isolation of Pfeiffer's bacillus" did not receive any particular attention.[57]
In May 1929, Fleming reported his findings to the British Journal of Experimental Pathology whch published them in the next month's issue.[71][78] It failed to attract any serious attention. Fleming himself was quite unsure of the medical application and was more concerned about the application for bacterial isolation, as he concluded:
In addition to its possible use in the treatment of bacterial infections penicillin is certainly useful to the bacteriologist for its power of inhibiting unwanted microbes in bacterial cultures so that penicillin insensitive bacteria can readily be isolated. A notable instance of this is the very easy, isolation of Pfeiffers bacillus of influenza when penicillin is used...It is suggested that it may be an efficient antiseptic for application to, or injection into, areas infected with penicillin-sensitive microbes.[71]
G. E. Breen, a fellow member of the Chelsea Arts Club, once asked Fleming: "I just wanted you to tell me whether you think it will ever be possible to make practical use of the stuff [penicillin]. For instance, could I use it?" Fleming gazed vacantly for a moment and then replied: "I don't know. It's too unstable. It will have to be purified, and I can't do that by myself."[57] Even as late as in 1941, the British Medical Journal reported that "the main facts emerging from a very comprehensive study [of penicillin] in which a large team of workers is engaged... does not appear to have been considered as possibly useful from any other point of view."[79][80][c]
Isolation
In 1933, Ernst Boris Chain, a chemist of Jewish-German origin, joined Australian scientist Howard Florey (later Baron Florey) at the Sir William Dunn School of Pathology at the University of Oxford in 1936 to investigate antibiotics.[81] Florey assigned him to work on lysozyme, an antibacterial enzyme discovered by Fleming in 1922.[82] In 1938, he came across Fleming's 1929 paper while writing a research report and informed his supervisor of the potential medical benefits of penicillin.[83] Although a year before, Florey had concentrated on pyocyanase (a pigment from the bacterium Bacillus pycyaneus, now called Pseudomonas aeruginosa), he agreed with Chain that penicillin was medically more promising.[84] In 1939, Florey and Chain obtained a research grant of $25,000 from the Rockefeller Foundation to study antibiotics,[85][86] which allowed them to assemble a research team composed of Edward Abraham, Arthur Duncan Gardner, Norman Heatley, Margaret Jennings, J. Orr-Ewing and G. Sanders.[87][88]
The Oxford team soon prepared a concentrated extract of P. rubens as "a brown powder" that "has been obtained which is freely soluble in water".[89] They found that the powder was not only effective in vitro against bacterial cultures but also and in vivo against bacterial infection in mice. On 5 May 1939, they injected a group of eight mice with a virulent strain of S. aureus, and then injected four of them with the penicillin solution. After one day, all the untreated mice died while the penicillin-treated mice survived; "a miracle" in Chain's view.[83] The team published its findings in The Lancet in 1940.[89]
The team reported details of the isolation method in 1941 with a scheme for large-scale extraction. They also found that penicillin was most abundant as a yellow concentrate from the mould extract,[90] but it was able to produce only small quantities. By the early 1942, they could prepare highly purified compound,[91] and derived the empirical chemical formula as C24H32O10N2Ba.[92] In the June 1942 issue of the British Journal of Experimental Pathology, Chain, Abraham and E. R. Holiday reported the production of the pure compound concluding that:
The penicillin preparation described in this paper is the most powerful antibacterial agent with predominantly bacteriostatic action so far known. Though it has not yet been obtained crystalline there are indications that it possesses a considerable degree of purity... The unusual biological properties of penicillin are linked with an exceptionally unstable chemical configuration. Inactivation by acid, alkali, and by boiling at any pH has been shown to be accompanied by definite chemical changes.[93]
First medical use
In January 1929, Fleming performed the first clinical trial with penicillin on his assistant Craddock. Craddock had developed a severe infection of the nasal antrum (sinusitis) for which he had undergone surgery. Fleming made use of the surgical opening of the nasal passage and started injecting penicillin on 9 January. Craddock showed no sign of improvement, probably because the infection was caused by influenza bacillus (Haemophilus influenzae), the bacterium which he had found not susceptible to penicillin.[94] It seems that Fleming gave some of his original penicillin samples to his colleague-surgeon Arthur Dickson Wright for clinical test in 1928.[95][96] Although Wright reportedly said that it: "seemed to work satisfactorily,"[23] there are no records of its specific use.
In November 1930, Cecil George Paine, a pathologist at the Royal Infirmary in Sheffield, was the first to use penicillin for medical treatment successfully.[15] He was a former student of Fleming who, after learning about penicillin, requested a sample from Fleming.[97] He initially attempted to treat sycosis (eruptions in beard follicles). On 25 November, he then tried it successfully with four patients (one adult, the others infants) who had ophthalmia neonatorum, an inflammation of the eye due to infection.[98] Thus, penicillin first worked on an eye infection.[99]
Nine years later, the Oxford team showed that Penicillium extract killed different bacteria (Streptococcus pyogenes, Staphylococcus aureus, and Clostridium septique) in culture and effectively cured Streptococcus infection in mice.[83] Thus, they reported their findings in the 24 August 1940 issue of The Lancet under the title "Penicillin as a chemotherapeutic agent" concluding:
The results are clear cut, and show that penicillin is active in vivo against at least three of the organisms inhibited in vitro. It would seem a reasonable hope that all organisms in high dilution in vitro will be found to be dealt with in vivo. Penicillin does not appear to be related to any chemotherapeutic substance at present in use and is particularly remarkable for its activity against the anaerobic organisms associated with gas gangrene.[89]
The following year, the Oxford team treated a policeman, Albert Alexander, who had a severe facial infection; his condition improved, but he eventually died as the researchers ran out of penicillin.[100] Subsequently, several other patients were treated successfully,[101] among them the survivors of the Cocoanut Grove fire in Boston (December 1942) who were the first burn patients to be successfully treated with penicillin.[102]
The most important clinical test took place in August 1942 when Fleming cured Harry Lambert (a work associate of Robert, Fleming's brother) of a fatal infection of the nervous system (streptococcal meningitis).[103] Fleming asked Florey for a purified penicillin sample, which he immediately injected into Lambert's spinal canal. Lambert showed signs of improvement the next day,[104] and completely recovered within a week.[105][106] Fleming reported his findings in The Lancet in 1943.[107] It was on this medical evidence that the British War Cabinet set up the Penicillin Committee on 5 April 1943 formed by Cecil Weir, Director General of Equipment, as Chairman, Fleming, Florey, Sir Percival Hartley, Allison and representatives from pharmaceutical companies as members.[104] The establishment of the committee opened the door to the mass production of penicillin the next year.[108][109]
Mass production
Crulina 98, CC-BY 3.0
Knowing that large-scale production for medical use was futile in a confined laboratory, the Oxford team tried to persuade the war-torn British government and private companies to undertake mass production, but in vain.[110] Florey and Heatley travelled to the United States (US) in June 1941 to persuade the American government and pharmaceutical companies there.[111] Ton ensure safe transportation of the sample, they smeared their coat pockets with the mould instead of taking it in a vial.[83] In July, they met with Andrew Jackson Moyer and Robert D. Coghill at the USDA Northern Regional Research Laboratory (NRRL, now the National Center for Agricultural Utilization Research) in Peoria, Illinois, where large-scale fermentations were done.[112] The Americans showed great interest and were able to make a Penicillium culture by the end of July[110] but realised that Fleming's mould was not efficient enough to produce large quantities of penicillin.[113]
With the help of US Army Transport Command, NRRL mycologist Kenneth Bryan Raper was able to locate similar but better moulds from Chungkin (China), Bombay (Mumbai, India) and Cape Town (South Africa). However, the single-best sample was obtained in 1943 from cantaloupe (a type of melon) sold in the Peoria fruit market. The mould was identified to be P. chrysogenum and designated as "NRRL 1951" or "cantaloupe strain" (Figure 6).[112][114] There is a popular story that Mary K. Hunt (or Mary Hunt Stevens[115]), a staff member at NRRL, collected the mould;[116] for which she was popularised as "Mouldy Mary."[117][118] However, Raper remarked that this story was "folklore" and that the fruit was delivered to the laboratory by a woman from the local fruit market.[112]
Between 1941 and 1943, Moyer, Coghill and Raper developed methods for industrialized penicillin production and isolated higher-yielding strains of the Penicillium mould.[119] Simultaneous research by Jasper H. Kane and other Pfizer scientists in Brooklyn developed the practical, deep-tank fermentation method for production of large quantities of pharmaceutical-grade penicillin.[120]

National Institute of Health, Public domain
When production first began, one-litre containers had a yield of less than 1%, but improved to a yield of 80–90% in 10,000 gallon containers.[88] This increase in efficiency happened between 1941 and 1945 as the result of continuous process innovation (Figure 7 shows one of the first mass applications).[121] Orvill May, director of the Agricultural Research Service, had Coghill use his experience with fermentation to increase the efficiency of extracting penicillin from the mould. When Moyer and Coghill replaced sucrose with lactose in the growth media, penicillin yield was increased.[122] An even larger increase occurred when they added corn steep liquor.[123]
The inefficiency of growing the mould on the surface of their nutrient baths, rather than having it submerged was a major challenge to the scientists. Although a submerged process of growing the mould was more efficient, the strain used was not suitable for the required conditions.[124] With the improved fermentation, the cantaloupe strain could produced six times as much penicillin per millilitre of the culture filtrate.[125] To improve on the cantaloupe strain, researchers subjected it to X-rays to facilitate mutations in its genome.[88] The new mutant strain, designated "X-1612", made in in 1946 produced twice as much penicillin as the original strain.[126] Another mutant strain, named "Q176", made in 1947 from ultraviolet irradiation further doubled the penicillin productivity.[127]
Now scientists had a mould that grew well submerged and produced an acceptable amount of penicillin. The next challenge was to provide the air required by the mould to grow. This problem was solved using an aerator but, due to the use of corn steep, aeration caused severe foaming.[128] The addition of an anti-foaming agents such as arachis oil, lard oil and other fatty acids solved this problem.[124][129] These anti-foaming agents further enhanced penicillin production.[130]
Chemical analysis
The chemical structure of penicillin was first proposed by Edward Abraham in 1942,[87] and three years later, Dorothy Hodgkin, working at Oxford, determined the correct chemical structure using X-ray crystallography.[131][132] The same year, chemical analyses done at different universities, pharmaceutical companies and government research departments was published jointly by the US Committee on Medical Research and the British Medical Research Council in the journal Science. The report announced the existence of different forms of penicillin compounds that shared the same structural component called β-lactam.[133] In the United Kingdom the penicillins were called penicillin I, II, III, and IV (Roman numerals were used according to the order of their discovery) while in the US scientists used letters such as F, G, K, and X that referred to their origins or sources as shown below:
| UK nomenclature | US nomenclature | Chemical name |
|---|---|---|
| Penicillin I | Penicillin F | 2-Pentenylpenicillin |
| Penicillin II | Penicillin G | Benzylpenicillin |
| Penicillin III | Penicillin X | p-Hydroxybenzylpenicillin |
| Penicillin IV | Penicillin K | n-Heptylpenicillin |
The use of two different names for each penicillin caused confusion.[134] As the chemical structures came to be known, the chemical names (based on the side chains of the compounds[135]) further complicated their identification and application. Thus, penicillin literature became a mixture of three naming systems. Chemists mostly adhered to the chemical names,[136][135] while biologists preferred the classic numbered or lettered names.[137][138] To resolve the confusion, in 1948, Chain introduced the chemical names as standard nomenclature, remarking: "To make the nomenclature as far as possible unambiguous it was decided to replace the system of numbers or letters by prefixes indicating the chemical nature of the side chain R."[139]
Further developments took place. In Austria, Hans Margreiter and Ernst Brandl of Biochemie (now Sandoz) developed the first acid-stable penicillin for oral administration, penicillin V in 1952.[140] American chemist John C. Sheehan at the Massachusetts Institute of Technology (MIT) completed the first chemical synthesis of penicillin in 1957.[141][142][143] Sheehan had started his studies into penicillin synthesis in 1948, and during these investigations developed new methods for the synthesis of peptides, as well as new protecting groups—groups that mask the reactivity of certain functional groups.[143][144] Although the initial synthesis developed by Sheehan was not appropriate for mass production of penicillins, one of the intermediate compounds in his synthesis was 6-aminopenicillanic acid (6-APA), the nucleus of penicillin.[145][146]
An important moment in the history of penicillin was the discovery of 6-APA itself. In 1957, researchers at Surrey's Beecham Research Laboratories (now the Beecham Group) isolated 6-APA from the culture media of P. chrysogenum. As published in Nature (1959),[147] 6-APA was found to constitute the core 'nucleus' of penicillin (in fact, all β-lactam antibiotics) and was easily chemically modified by attaching side chains through chemical reactions.[148][149] This discovery paved the way for new and improved drugs as all semi-synthetic penicillins are produced from chemical manipulation of 6-APA.[150]
The second-generation semi-synthetic β-lactam antibiotic methicillin, designed to counter first-generation-resistant penicillinases, was introduced in the United Kingdom in 1959. It is likely that methicillin-resistant forms of Staphylococcus aureus already existed at the time.[151][152]
Outcomes
Penicillin patents became a matter of concern and conflict. Chain had wanted to apply for a patent but Florey and his teammates had objected arguing that penicillin should benefit all.[153] He sought the advice of Sir Henry Hallett Dale (Chairman of the Wellcome Trust and member of the Scientific Advisory Panel to the Cabinet of British government) and John William Trevan (Director of the Wellcome Trust Research Laboratory). On 26 and 27 March 1941, Dale and Trevan met at Sir William Dunn School of Pathology to discuss the issue. Dale specifically advised that patenting penicillin would be unethical.[154] Not giving up, Chain approached Sir Edward Mellanby, then Secretary of the Medical Research Council, who also objected on ethical grounds.[155] As Chain later admitted, he had "many bitter fights" with Mellanby,[154] but Mellanby's decision was accepted as final.[155]
In 1945, Moyer patented the methods for production and isolation of penicillin.[156][157][158] Moyer could not obtain a patent in the US as an employee of the NRRL, and filed his patent at the British Patent Office (now the Intellectual Property Office). He gave the license to a US company, Commercial Solvents Corporation. Although completely legal, his colleague Coghill felt it was an injustice for outsiders to have the royalties for the "British discovery." A year later, Moyer asked Coghill for permission to file another patent based on the use of phenylacetic acid that increased penicillin production by 66%, but as the principal researcher, Coghill refused.[159]
When Fleming learned of the American patents on penicillin production, he was infuriated and commented:
I found penicillin and have given it free for the benefit of humanity. Why should it become a profit-making monopoly of manufacturers in another country?[160]
Fleming, Florey and Chain shared the 1945 Nobel Prize in Physiology or Medicine "for the discovery of penicillin and its curative effect in various infectious diseases."[161] Hodgkin received the 1964 Nobel Prize in Chemistry "for her determinations by X-ray techniques of the structures of important biochemical substances."[162]
Development of penicillin-derivatives
The narrow range of treatable diseases of the penicillins prompted further search for derivatives of penicillin. The isolation of 6-APA allowed preparation of semisynthetic penicillins, with various improvements in terms of bioavailability, spectrum, stability, tolerance.[163] The first major development was the production of ampicillin by the Beecham Research Laboratories in London in 1961.[164] Ampicillin had advantages over the original penicillin as it showed activity against both Gram-positive and Gram-negative bacteria.[164] Further development yielded β-lactamase-resistant penicillins, including flucloxacillin, dicloxacillin, and methicillin, which were specifically active against β-lactamase-producing bacterial species, but not against the methicillin-resistant Staphylococcus aureus strains that subsequently emerged.[165]
Another development in penicillin synthesis was the antipseudomonal penicillins, such as carbenicillin, ticarcillin, and piperacillin, useful for their activity against Gram-negative bacteria. The penicillins and related β-lactams have become the most widely used antibiotics in the world.[166] Amoxicillin, a semisynthetic penicillin developed by Beecham Research Laboratories in 1970,[167][168] is the single-most commonly used.[169][170] However, the usefulness of these penicillins and later developed antibiotics, including the mecillinams, the carbapenems and, most important, the cephalosporins, is limited by the common structure, the β-lactam ring,[149][171] which is not only the antibiotic active part of the chemical compound, but also the target site of bacteria in drug resistance.[172]
Drug resistance
In his Nobel lecture, Fleming warned of the possibility of penicillin resistance in clinical conditions:
The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily underdose himself and by exposing his microbes to non-lethal quantities of the drug make them resistant.[66]
In 1940, Chain and Abraham reported the first indication of antibiotic resistance to penicillin, an E. coli strain that produced the penicillinase enzyme, which was capable of breaking down penicillin and completely negating its antibacterial effect.[151][78] They worked out the chemical nature of penicillinase which they reported in Nature:
The conclusion that the active substance is an enzyme is drawn from the fact that it is destroyed by heating at 90° for 5 minutes and by incubation with papain activated with potassium cyanide at pH 6, and that it is non-dialysable through 'Cellophane' membranes.[173]
By 1942, some strains of Staphylococcus aureus had developed a strong resistance to penicillin. Eighteen years later, most of the strains were resistant to penicillin.[174] In 1967, Streptococcus pneumoniae was also reported to be penicillin resistant. Many other strains of bacteria have eventually developed, and continue to develop a resistance to penicillin.[175][78]
Additional information
Acknowledgement
Literature access provided by the Wikipedia Library.
Competing interests
The authors have no competing interests.
Ethics statement
No ethics approval applicable.
Funding
None.
Notes
- ↑ Unlike his predecessor scientists such as Francesco Redi and Louis Pasteur who had experimentally rebutted spontaneous generation, Burdon-Sanderson went further by specifically identifying the types of microbes as bacteria, microzyme, fungi, torula, and Penicillium, including the differences in their growth pattern. His experiment on 11 November 1870 reads: "At the same date all the glasses showed tufts of penicillium; those on 3 and 5 were more advanced than the rest... the liquid in 5 was found to be perfectly limpid and free from microzymes (referring to bacteria)."
- ↑ At the time, the term Penicillium glaucum was used as a catch-all phrase for a variety of different fungi, though not for Penicillium notatum. Duchesne's specific mold was unfortunately not preserved, which makes it impossible to be certain today which fungus might have been responsible for the cure and, consequently, even less certain which specific antibacterial substance was responsible.
- ↑ The statement "does not appear to have been considered as possibly useful from any other point of view" seems to be later deleted, but is still apparent from Fleming's response (BMJ, 1941, 2 (4210): 386–386).
References
- ↑ Aminov, Rustam I. (2010). "A brief history of the antibiotic era: lessons learned and challenges for the future". Frontiers in Microbiology 1: 134. doi:10.3389/fmicb.2010.00134. PMID 21687759. PMC 3109405. https://pubmed.ncbi.nlm.nih.gov/21687759.
- ↑ Hutchings, Matthew I.; Truman, Andrew W.; Wilkinson, Barrie (2019). "Antibiotics: past, present and future". Current Opinion in Microbiology 51: 72–80. doi:10.1016/j.mib.2019.10.008. PMID 31733401. https://pubmed.ncbi.nlm.nih.gov/31733401.
- ↑ Ehrlich, Paul; Hata, S. (1910). Die experimentelle Chemotherapie der Spirillosen (in German). Berlin, Heidelberg: Springer Berlin Heidelberg. pp. 1-178. doi:10.1007/978-3-642-64926-4. ISBN 978-3-642-64911-0. http://link.springer.com/10.1007/978-3-642-64926-4.
- ↑ Williams, K. J. (2009). "The introduction of 'chemotherapy' using arsphenamine - the first magic bullet". Journal of the Royal Society of Medicine 102 (8): 343–348. doi:10.1258/jrsm.2009.09k036. PMID 19679737. PMC 2726818. https://pubmed.ncbi.nlm.nih.gov/19679737.
- ↑ Swain, K. (2018). "'Extraordinarily arduous and fraught with danger': syphilis, Salvarsan, and general paresis of the insane". The Lancet Psychiatry 5 (9): 702–703. doi:10.1016/S2215-0366(18)30221-9. PMID 29866584. https://pubmed.ncbi.nlm.nih.gov/29866584.
- ↑ Park, J. T.; Strominger, J. L. (1957). "Mode of action of penicillin". Science 125 (3238): 99–101. doi:10.1126/science.125.3238.99. PMID 13390969. https://pubmed.ncbi.nlm.nih.gov/13390969.
- ↑ Clement, Meredith E.; Okeke, N. Lance; Hicks, Charles B. (2014-11-12). "Treatment of syphilis: a systematic review". JAMA 312 (18): 1905–1917. doi:10.1001/jama.2014.13259. ISSN 1538-3598. PMID 25387188. PMC 6690208. https://pubmed.ncbi.nlm.nih.gov/25387188.
- ↑ Ji, Hong-Fang; Li, Xue-Juan; Zhang, Hong-Yu (2009). "Natural products and drug discovery. Can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia?". EMBO Reports 10 (3): 194–200. doi:10.1038/embor.2009.12. PMID 19229284. PMC 2658564. https://pubmed.ncbi.nlm.nih.gov/19229284.
- ↑ "History of Antibiotics | Steps of the Scientific Method, Research and Experiments". Experiment-Resources.com. Archived from the original on 6 August 2011. Retrieved 2012-07-13.
- ↑ Wainwright, Milton (1989). "Moulds in Folk Medicine". Folklore 100 (2): 162–166. doi:10.1080/0015587X.1989.9715763. https://www.tandfonline.com/doi/pdf/10.1080/0015587X.1989.9715763.
- ↑ Cranch, A. G. (1943). "Early use of Penicillin (?)". Journal of the American Medical Association 123 (15): 990. doi:10.1001/jama.1943.02840500054025. ISSN 0002-9955. http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.1943.02840500054025.
- ↑ Gould, Kate (2016). "Antibiotics: from prehistory to the present day". The Journal of Antimicrobial Chemotherapy 71 (3): 572–575. doi:10.1093/jac/dkv484. PMID 26851273. https://pubmed.ncbi.nlm.nih.gov/26851273/.
- ↑ Wainwright, Milton; Rally, Louise; Ali, Tasneem Adam (1992). "The scientific basis of mould therapy". Mycologist 6 (3): 108–110. doi:10.1016/S0269-915X(09)80587-9. https://linkinghub.elsevier.com/retrieve/pii/S0269915X09805879.
- ↑ Singh, Jagjit (1999). Singh, Jagjit. ed. From Ethnomycology to Fungal Biotechnology (in en). Boston, MA: Springer US. pp. 11–17. doi:10.1007/978-1-4615-4815-7_2. ISBN 978-1-4613-7182-3. http://link.springer.com/10.1007/978-1-4615-4815-7_2.
- ↑ 15.0 15.1 Wainwright, Milton (1989). "Moulds in Folk Medicine". Folklore 100 (2): 162–166. doi:10.1080/0015587X.1989.9715763. http://www.tandfonline.com/doi/abs/10.1080/0015587X.1989.9715763.
- ↑ Wainwright, Milton (1989). "Moulds in ancient and more recent medicine". Mycologist 3 (1): 21–23. doi:10.1016/S0269-915X(89)80010-2. https://linkinghub.elsevier.com/retrieve/pii/S0269915X89800102.
- ↑ Hutchings, Matthew I.; Truman, Andrew W.; Wilkinson, Barrie (2019). "Antibiotics: past, present and future". Current Opinion in Microbiology 51: 72–80. doi:10.1016/j.mib.2019.10.008. PMID 31733401. https://pubmed.ncbi.nlm.nih.gov/31733401.
- ↑ Queener, Sherry; Webber, J. Alan; Queener, Stephen; eds. (1986). Beta-lactam Antibiotics for Clinical Use, Informa Health Care, ISBN 0824773861, p. 4. [Refers to : J. B. Sanderson. Appendix No 5. " Further report of researches concerning the intimate pathology of contagion. The origin and distribution of microzymes (bacteria) in water, and the circumstances which determine their existence in the tissue and liquids of the living body ". 13th Report of the Medical Officer of the Privy Council [John Simon], with Appendix, 1870. Her Majesty's Stationery Office, London, 1871, pp. 56–66; reprinted in Quarterly Journal of Microscopical Science, n. ser., XI, 1871, pp. 323–352.]
- ↑ Burdon-Sanderson, (John) (1871). "Memoirs: The Origin and Distribution of Microzymes (Bacteria) in Water, and the Circumstances which determine their Existence in the Tissues and Liquids of the Living Body". Journal of Cell Science s2-11 (44): 323–352. doi:10.1242/jcs.s2-11.44.323. https://doi.org/10.1242/jcs.s2-11.44.323.
- ↑ 20.0 20.1 20.2 Selwyn, S. (1979). "Pioneer work on the ‘penicillin phenomenon’, 1870–1876". Journal of Antimicrobial Chemotherapy 5 (3): 249–255. doi:10.1093/jac/5.3.249. https://academic.oup.com/jac/article-lookup/doi/10.1093/jac/5.3.249.
- ↑ Majno, Guido; Joris, Isabelle (1979). "Billroth and Penicillium". Reviews of Infectious Diseases 1 (5): 880–884. doi:10.1093/clinids/1.5.880. https://www.jstor.org/stable/4452384.
- ↑ Lister, Joseph (1875). "XVI.— A Contribution to the Germ Theory of Putrefaction and other Fermentative Changes, and to the Natural History of Torulæ and Bacteria". Transactions of the Royal Society of Edinburgh 27 (3): 313–344. doi:10.1017/S0080456800026004. https://www.cambridge.org/core/product/identifier/S0080456800026004/type/journal_article.
- ↑ 23.0 23.1 Wainwright, M (1987). "The history of the therapeutic use of crude penicillin.". Medical History 31 (1): 41–50. doi:10.1017/s0025727300046305. PMID 3543562. PMC 1139683. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1139683/.
- ↑ Foster, W.; Raoult, A. (1974). "Early descriptions of antibiosis". The Journal of the Royal College of General Practitioners 24 (149): 889–894. PMID 4618289. PMC 2157443. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2157443/.
- ↑ Roberts, W. (1874). "Studies on biogenesis". Philosophical Transactions of the Royal Society of London 164: 457–477. doi:10.1098/rstl.1874.0012. https://www.jstor.org/stable/109106.
- ↑ Doetsch, R. N. (1963). "Studies on biogenesis by Sir William Roberts". Medical History 7: 232–240. doi:10.1017/s0025727300028374. PMID 14028366. PMC 1034828. https://pubmed.ncbi.nlm.nih.gov/14028366.
- ↑ Landsberg, H. (1949). "Prelude to the discovery of penicillin". Isis 40 (3): 225–227. doi:10.1086/349043. https://www.jstor.org/stable/227238.
- ↑ Koch, Robert (2010). Robert Koch-Institut. "Die Ätiologie der Milzbrand-Krankheit, begründet auf die Entwicklungsgeschichte des Bacillus Anthracis". Cohns Beiträge zur Biologie der Pflanzen 2 (2): 277 (1–22). doi:10.25646/5064. https://edoc.rki.de/handle/176904/5139.
- ↑ Lakhtakia, Ritu (2014). "The Legacy of Robert Koch: Surmise, search, substantiate". Sultan Qaboos University Medical Journal 14 (1): e37–41. doi:10.12816/0003334. PMID 24516751. PMC 3916274. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3916274/.
- ↑ 30.0 30.1 30.2 Sharma, G. (2016). "La Moisissure et la Bactérie: Deconstructing the fable of the discovery of penicillin by Ernest Duchesne". Endeavour 40 (3): 188–200. doi:10.1016/j.endeavour.2016.07.005. PMID 27496372. https://dspace.lboro.ac.uk/2134/21810.
- ↑ 31.0 31.1 Florey, Howard W. (1946). "The Use of Micro-organisms for Therapeutic Purposes". The Yale Journal of Biology and Medicine 19 (1): 101–118.1. PMID 20275724. PMC 2602034. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2602034/.
- ↑ Foster, W.; Raoult, A. (1974). "Early descriptions of antibiosis". The Journal of the Royal College of General Practitioners 24 (149): 889–894. PMID 4618289. PMC 2157443. https://pubmed.ncbi.nlm.nih.gov/4618289.
- ↑ 33.0 33.1 Brunel, J. (1951). "Antibiosis from Pasteur to Fleming". Journal of the History of Medicine and Allied Sciences 6 (3): 287–301. doi:10.1093/jhmas/vi.summer.287. PMID 14873929. https://pubmed.ncbi.nlm.nih.gov/14873929.
- ↑ Waksman, S. A. (1947). "What is an antibiotic or an antibiotic substance?". Mycologia 39 (5): 565–569. doi:10.1080/00275514.1947.12017635. PMID 20264541. https://pubmed.ncbi.nlm.nih.gov/20264541.
- ↑ Kruif, Paul De (1996). Microbe Hunters (in en). Florida (USA): Houghton Mifflin Harcourt. pp. 144. ISBN 978-0-15-602777-9. https://books.google.co.in/books?redir_esc=y&id=pH24vLpivRgC&q. "At once Pasteur jumped to a fine idea: "If the harmless bugs from the air choke out the anthrax bacilli in the bottle, they will do it in the body too! It is a kind of dog-eat-dog!” shouted Pasteur, (...) Pasteur gravely announced: "That there were high hopes for the cure of disease from this experiment", but that is the last you hear of it, for Pasteur was never a man to give the world of science the benefit of studying his failures."
- ↑ Tiberio, Vincenzo (1895) "Sugli estratti di alcune muffe" [On the extracts of certain moulds], Annali d'Igiene Sperimentale (Annals of Experimental Hygiene), 2nd series, 5 : 91–103. From p. 95: "Risulta chiaro da queste osservazioni che nella sostanza cellulare delle muffe esaminate son contenuti dei principi solubili in acqua, forniti di azione battericida: sotto questo riguardo sono più attivi o in maggior copia quelli dell' Asp. flavescens, meno quelli del Mu. mucedo e del Penn. glaucum." (It follows clearly from these observations that in the cellular substance of the moulds examined are contained some water-soluble substances, provided with bactericidal action: in this respect are more active or in greater abundance those of Aspergillus flavescens; less, those of Mucor mucedo and Penicillium glaucum.)
- ↑ 37.0 37.1 Perciaccante, Antonio; Coralli, Alessia; Lippi, Donatella; Appenzeller, Otto; Bianucci, Raffaella (2019). "Vincenzo Tiberio (1869-1915) and the dawn of the antibiotic age". Internal and Emergency Medicine 14 (8): 1363–1364. doi:10.1007/s11739-019-02116-1. PMID 31154612. https://pubmed.ncbi.nlm.nih.gov/31154612.
- ↑ 38.0 38.1 Bucci, Roberto; Galli, P. (2011). "Vincenzo Tiberio: a misunderstood researcher". Italian Journal of Public Health 8 (4): 404–406. https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.897.8253&rep=rep1&type=pdf.
- ↑ Kyle, R. A. (2005). "Five decades of therapy for multiple myeloma: a paradigm for therapeutic models". Leukemia 19 (6): 910–912. doi:10.1038/sj.leu.2403728. PMID 15800669. https://pubmed.ncbi.nlm.nih.gov/15800669.
- ↑ 40.0 40.1 Lehrer, Steven (2006). Explorers of the Body: Dramatic Breakthroughs in Medicine from Ancient Times to Modern Science (in en) (2 ed.). Lincoln, Nebraska: iUniverse. pp. 331-332. ISBN 9780595407316. https://books.google.com/books?id=cIZpyNvjvdcC&newbks=0&hl.
- ↑ Duchesne, E (1897). Contribution à l'étude de la concurrence vitale chez les micro-organismes : antagonisme entre les moisissures et les microbes (in fr). Lyon, France: Alexandre Rey. https://books.google.com/books?id=5L8UAAAAYAAJ&pg=PA1.
- ↑ Pouillard, Jean. "Une découverte oubliée : la thèse de médecine du docteur Ernest Duchesne (1874–1912)". Histoire des Sciences Médicales XXXVI (1): 11–20. Archived from the original on 13 July 2019. http://www.biusante.parisdescartes.fr/sfhm/hsm/HSMx2002x036x001/HSMx2002x036x001x0011.pdf.
- ↑ 43.0 43.1 Schaefer, B. (2015). Natural Products in the Chemical Industry. Springer. p. 231. doi:10.1007/978-3-642-54461-3. ISBN 9783642544613. https://books.google.com/books?id=DbO4CQAAQBAJ&pg=PA231.
- ↑ Duckett, S. (1999). "Ernest Duchesne and the concept of fungal antibiotic therapy". The Lancet 354 (9195): 2068–2071. doi:10.1016/S0140-6736(99)03162-1. https://linkinghub.elsevier.com/retrieve/pii/S0140673699031621.
- ↑ Luengo, José M. (1999). "Enzymatic Synthesis of Penicillins". In Barton, D.. Comprehensive Natural Products Chemistry (in en). Elsevier. pp. 239–274. doi:10.1016/b978-0-08-091283-7.00105-3. ISBN 978-0-08-091283-7. https://linkinghub.elsevier.com/retrieve/pii/B9780080912837001053.
- ↑ 46.0 46.1 Peters, Johan T. (2009). "The First Discoverers of Penicillin and of its Application in Therapy". Acta Medica Scandinavica 126 (1): 60–64. doi:10.1111/j.0954-6820.1946.tb19000.x. http://doi.wiley.com/10.1111/j.0954-6820.1946.tb19000.x.
- ↑ Elvehjem, C. A. (1940). "Relation of nicotinic acid to pellagra". Physiological Reviews 20 (2): 249–271. doi:10.1152/physrev.1940.20.2.249. https://www.physiology.org/doi/10.1152/physrev.1940.20.2.249.
- ↑ Sydenstricker, V. P. (1958). "The history of pellagra, its recognition as a disorder of nutrition and its conquest". The American Journal of Clinical Nutrition 6 (4): 409–414. doi:10.1093/ajcn/6.4.409. PMID 13559167. https://pubmed.ncbi.nlm.nih.gov/13559167.
- ↑ 49.0 49.1 Mohr, Kathrin I. (2016). "History of Antibiotics Research". In Stadler, Marc. How to Overcome the Antibiotic Crisis. 398. Cham: Springer International Publishing. pp. 237–272. doi:10.1007/82_2016_499. ISBN 978-3-319-49282-7. http://link.springer.com/10.1007/82_2016_499.
- ↑ Gosio, Bartolomeo (1896). Ricerche batteriologiche e chimiche sulle alterazioni del mais: contributo all'etiologia della pellagra (in it). Tip. delle mantellate. https://books.google.com/books?id=GTXooAEACAAJ&newbks=0&hl=en.
- ↑ Nicolaou, Kyriacos C.; Rigol, Stephan (2018). "A brief history of antibiotics and select advances in their synthesis". The Journal of Antibiotics 71 (2): 153–184. doi:10.1038/ja.2017.62. PMID 28676714. https://pubmed.ncbi.nlm.nih.gov/28676714.
- ↑ Halle, Mahesh B.; Lee, Woohyun; Yudhistira, Tesla; Kim, Myungseob; Churchill, David G. (2019). "Mycophenolic Acid: Biogenesis, Compound Isolation, Biological Activity, and Historical Advances in Total Synthesis". European Journal of Organic Chemistry 2019 (13): 2315–2334. doi:10.1002/ejoc.201900245. http://doi.wiley.com/10.1002/ejoc.201900245.
- ↑ 53.0 53.1 Wainwright, Milton (2000). "André Gratia (1893–1950): Forgotten Pioneer of Research into Antimicrobial Agents". Journal of Medical Biography 8 (1): 39–42. doi:10.1177/096777200000800108. PMID 11608911. http://journals.sagepub.com/doi/10.1177/096777200000800108.
- ↑ de Scoville, C; Brouwer, C De; Dujardin, M (1999). "Nobel chronicle: Fleming and Gratia". The Lancet 354 (9174): 258. doi:10.1016/S0140-6736(05)66334-9. PMID 10421340. https://linkinghub.elsevier.com/retrieve/pii/S0140673605663349.
- ↑ Fraser, I. (1984). "Penicillin: early trials in war casualties". British Medical Journal 289 (6460): 1723–1725. doi:10.1136/bmj.289.6460.1723. PMID 6440621. PMC 1444789. https://pubmed.ncbi.nlm.nih.gov/6440621.
- ↑ Eickhoff, Theodore C. (2008). "Penicillin: An accidental discovery changed the course of medicine". Endocrine Today. Retrieved 2021-10-10.
- ↑ 57.0 57.1 57.2 57.3 57.4 57.5 57.6 Lalchhandama, K. (2020). "Reappraising Fleming's snot and mould". Science Vision 20 (1): 29–42. doi:10.33493/scivis.20.01.03.
- ↑ Bigger, Joseph W.; Boland, C. R.; O'meara, R. A. Q. (1927). "Variant colonies ofStaphylococcus aureus". The Journal of Pathology and Bacteriology 30 (2): 261–269. doi:10.1002/path.1700300204. https://onlinelibrary.wiley.com/doi/10.1002/path.1700300204.
- ↑ 59.0 59.1 59.2 59.3 59.4 Diggins, F.W. (1999). "The true history of the discovery of penicillin, with refutation of the misinformation in the literature". British Journal of Biomedical Science 56 (2): 83–93. PMID 10695047.
- ↑ 60.0 60.1 60.2 60.3 Hare, R. (1982). "New light on the history of penicillin". Medical History 26 (1): 1–24. doi:10.1017/S0025727300040758. PMID 7047933. PMC 1139110. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1139110/.
- ↑ 61.0 61.1 61.2 Wyn Jones, Emyr; Wyn Jones, R. Gareth (2002). "Merlin Pryce (1902-1976) and penicillin: an abiding mystery". Vesalius 8 (2): 6–25. PMID 12713008. https://pubmed.ncbi.nlm.nih.gov/12713008.
- ↑ 62.0 62.1 Wainright, M. (1993). "The mystery of the plate: Fleming's discovery and contribution to the early development of penicillin". Journal of Medical Biography 1 (1): 59–65. doi:10.1177/096777209300100113. PMID 11639213.
- ↑ Tan, Siang Yong; Tatsumura, Yvonne (2015). "Alexander Fleming (1881-1955): Discoverer of penicillin". Singapore Medical Journal 56 (7): 366–367. doi:10.11622/smedj.2015105. PMID 26243971. PMC 4520913. https://pubmed.ncbi.nlm.nih.gov/26243971.
- ↑ Arseculeratne, S. N.; Arseculeratne, G. (2017). "A re-appraisal of the conventional history of antibiosis and Penicillin". Mycoses 60 (5): 343–347. doi:10.1111/myc.12599. PMID 28144986. https://pubmed.ncbi.nlm.nih.gov/28144986.
- ↑ Fleming, Alexander (1929). "On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzae". British Journal of Experimental Pathology 10 (3): 226–236. PMID 2048009. PMC 2041430. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2041430/.; Reprinted as "On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzae". British Journal of Experimental Pathology 60 (1): 3–13. 1979. PMC 2041430. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2041430/.
- ↑ 66.0 66.1 Fleming, Alexander (1999). "Penicillin: Nobel Lecture, December 11, 1945". Nobel Lectures, Physiology or Medicine, 1942-1962 (in en). Singapore: World Scientific. pp. 83–93. ISBN 978-981-02-3411-9. https://www.nobelprize.org/uploads/2018/06/fleming-lecture.pdf.
- ↑ Hess, Kristin (2019). "Fleming vs. Florey: It All Comes Down to the Mold". The Histories 2 (1): 3–10. https://digitalcommons.lasalle.edu/the_histories/vol2/iss1/3.
- ↑ Curry, J. (1981). "Obituary: C. J. La Touche". Sabouraudia 19 (2): 164. doi:10.1080/00362178185380261.
- ↑ Kingston, W. (2008). "Irish contributions to the origins of antibiotics". Irish Journal of Medical Science 177 (2): 87–92. doi:10.1007/s11845-008-0139-x. PMID 18347757. https://pubmed.ncbi.nlm.nih.gov/18347757.
- ↑ Henderson, J. W. (1997). "The yellow brick road to penicillin: a story of serendipity". Mayo Clinic Proceedings 72 (7): 683–687. doi:10.1016/S0025-6196(11)63577-5. PMID 9212774. https://pubmed.ncbi.nlm.nih.gov/9212774.
- ↑ 71.0 71.1 71.2 Fleming, Alexander (1929). "On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzae". British Journal of Experimental Pathology 10 (3): 226–236. PMID 2048009. PMC 2041430. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2041430/.; Reprint of "On the antibacterial action of cultures of a Penicillium, with special reference to their use in the isolation of B. influenzae". British Journal of Experimental Pathology 60 (1): 3–13. 1979. PMC 2041430. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2041430/.
- ↑ "Appendix. History of species used and Dr. Thom's diagnoses of species". Philosophical Transactions of the Royal Society of London. Series B, Containing Papers of a Biological Character 220 (468–473): 83–92. 1931. doi:10.1098/rstb.1931.0015.
- ↑ Thom, Charles (1945). "Mycology Presents Penicillin". Mycologia 37 (4): 460–475. doi:10.2307/3754632.
- ↑ Samson, R. A.; Hadlok, R.; Stolk, A. C. (1977). "A taxonomic study of the Penicillium chrysogenum series". Antonie Van Leeuwenhoek 43 (2): 169–175. doi:10.1007/BF00395671. PMID 413477. https://pubmed.ncbi.nlm.nih.gov/413477.
- ↑ "International Code of Botanical Nomenclature (VIENNA CODE). Appendix IV Nomina specifica conservanda et rejicienda. B. Fungi". International Association of Plant Taxonomy. 2006. Retrieved 17 June 2020.
- ↑ Houbraken, Jos; Frisvad, Jens C.; Samson, Robert A. (2011). "Fleming's penicillin producing strain is not Penicillium chrysogenum but P. rubens". IMA fungus 2 (1): 87–95. doi:10.5598/imafungus.2011.02.01.12. PMID 22679592. PMC 3317369. https://pubmed.ncbi.nlm.nih.gov/22679592.
- ↑ Houbraken, J.; Frisvad, J. C.; Seifert, K. A.; Overy, D. P.; Tuthill, D. M.; Valdez, J. G.; Samson, R. A. (2012). "New penicillin-producing Penicillium species and an overview of section Chrysogena". Persoonia 29: 78–100. doi:10.3767/003158512X660571. PMID 23606767. PMC 3589797. https://pubmed.ncbi.nlm.nih.gov/23606767.
- ↑ 78.0 78.1 78.2 Lobanovska, M.; Pilla, G. (2017). "Penicillin's Discovery and Antibiotic Resistance: Lessons for the Future?". The Yale Journal of Biology and Medicine 90 (1): 135–145. PMID 28356901. PMC 5369031. //www.ncbi.nlm.nih.gov/pmc/articles/PMC5369031/.
- ↑ "Annotations". British Medical Journal 2 (4208): 310–2. August 1941. doi:10.1136/bmj.2.4208.310. PMID 20783842. PMC 2162429. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2162429/.
- ↑ Fleming, A. (1941). "Penicillin". British Medical Journal 2 (4210): 386. doi:10.1136/bmj.2.4210.386. PMC 2162878. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2162878/.
- ↑ Chain, E. (1972). "Thirty years of penicillin therapy". Journal of the Royal College of Physicians of London 6 (2): 103–131. doi:10.1098/rspb.1971.0098. PMID 4551373. PMC 5366029. https://pubmed.ncbi.nlm.nih.gov/4551373.
- ↑ Fleming, A (1922). "On a remarkable bacteriolytic element found in tissues and secretions". Proceedings of the Royal Society B 93 (653): 306–317. doi:10.1098/rspb.1922.0023.
- ↑ 83.0 83.1 83.2 83.3 Gaynes, Robert (2017). "The Discovery of Penicillin—New Insights After More Than 75 Years of Clinical Use". Emerging Infectious Diseases 23 (5): 849–853. doi:10.3201/eid2305.161556. PMC 5403050. http://wwwnc.cdc.gov/eid/article/23/5/16-1556_article.htm.
- ↑ Chain, E.; Florey, H. W. (1944). "The discovery of the chemotherapeutic properties of penicillin". British Medical Bulletin 2 (1): 5–7. doi:10.1093/oxfordjournals.bmb.a071033. https://academic.oup.com/bmb/article-lookup/doi/10.1093/oxfordjournals.bmb.a071033.
- ↑ Florey, H. W.; Abraham, E. P. (1951). "The work on penicillin at Oxford". Journal of the History of Medicine and Allied Sciences 6 (3): 302–317. doi:10.1093/jhmas/vi.summer.302. https://www.jstor.org/stable/24619871.
- ↑ Kong, Kok-Fai; Schneper, Lisa; Mathee, Kalai (2010). "Beta-lactam antibiotics: from antibiosis to resistance and bacteriology". Acta Pathologica, Microbiologica, et Immunologica Scandinavica 118 (1): 1–36. doi:10.1111/j.1600-0463.2009.02563.x. PMID 20041868. PMC 2894812. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2894812/.
- ↑ 87.0 87.1 Jones, David S.; Jones, John H. (2014-12-01). "Sir Edward Penley Abraham CBE. 10 June 1913 – 9 May 1999". Biographical Memoirs of Fellows of the Royal Society 60: 5–22. doi:10.1098/rsbm.2014.0002. ISSN 0080-4606. http://rsbm.royalsocietypublishing.org/content/60/5.1.
- ↑ 88.0 88.1 88.2 "Discovery and Development of Penicillin". International Historic Chemical Landmarks. American Chemical Society. Retrieved 21 August 2021.
- ↑ 89.0 89.1 89.2 Chain, E.; Florey, H. W.; Adelaide, M. B.; Gardner, A. D.; Heatley, N. G.; Jennings, M. A.; Orr-Ewing, J.; Sanders, A. G. (1940). "Penicillin as a chemotherapeutic agent". The Lancet 236 (6104): 226–228. doi:10.1016/S0140-6736(01)08728-1. PMID 8403666. https://pubmed.ncbi.nlm.nih.gov/8403666.
- ↑ Abraham, E. P.; Chain, E.; Fletcher, C. M.; Florey, H. W.; Gardner, A. D.; Heatley, N. G.; Jennings, M. A. (1992). "Further observations on penicillin. 1941". European Journal of Clinical Pharmacology 42 (1): 3–9. PMID 1541313. https://pubmed.ncbi.nlm.nih.gov/1541313.
- ↑ Abraham, E. P.; Chain, E. (1942). "Purification of Penicillin". Nature 149 (3777): 328. doi:10.1038/149328b0. http://www.nature.com/articles/149328b0.
- ↑ Abraham, E. P.; Baker, W.; Chain, E.; Florey, H. W.; Holiday, E. R.; Robinson, R. (1942). "Nitrogenous Character of Penicillin". Nature 149 (3778): 356. doi:10.1038/149356a0. http://www.nature.com/articles/149356a0.
- ↑ Abraham, E. P.; Chain, E.; Holiday, E. R. (1942). "Purification and Some Physical and Chemical Properties of Penicillin". British Journal of Experimental Pathology 23 (3): 103–119. PMC 2065494. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2065494/.
- ↑ Hare, R. (1982). "New light on the history of penicillin". Medical History 26 (1): 1–24. doi:10.1017/s0025727300040758. PMID 7047933. PMC 1139110. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1139110/.
- ↑ Wainwright, M.; Swan, H.T. (1987). "The Sheffield penicillin story". Mycologist 1 (1): 28–30. doi:10.1016/S0269-915X(87)80022-8. https://linkinghub.elsevier.com/retrieve/pii/S0269915X87800228.
- ↑ Wainwright, Milton (1990). "Besredka's "antivirus" in relation to Fleming's initial views on the nature of penicillin". Medical History 34 (1): 79–85. doi:10.1017/S0025727300050286. PMID 2405221. PMC 1036002. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1036002/.
- ↑ "Dr Cecil George Paine - Unsung Medical Heroes - Blackwell's Bookshop Online". blackwells.co.uk. Retrieved 2020-10-19.
- ↑ Wainwright, M.; Swan, H.T. (1986). "C.G. Paine and the earliest surviving clinical records of penicillin therapy". Medical History 30 (1): 42–56. doi:10.1017/S0025727300045026. PMID 3511336. PMC 1139580. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1139580/.
- ↑ Alharbi, Sulaiman Ali; Wainwright, Milton; Alahmadi, Tahani Awad; Salleeh, Hashim Bin; Faden, Asmaa A.; Chinnathambi, Arunachalam (2014). "What if Fleming had not discovered penicillin?". Saudi Journal of Biological Sciences 21 (4): 289–293. doi:10.1016/j.sjbs.2013.12.007. PMID 25183937. PMC 4150221. //www.ncbi.nlm.nih.gov/pmc/articles/PMC4150221/.
- ↑ Hamdy, Ronald C. (2006). "Penicillin is 65 years old!". Southern Medical Journal 99 (2): 192–193. doi:10.1097/01.smj.0000194524.83293.0d. PMID 16509564. https://pubmed.ncbi.nlm.nih.gov/16509564/.
- ↑ "Making Penicillin Possible: Norman Heatley Remembers". ScienceWatch. Thomson Scientific. 2007. Archived from the original on February 21, 2007. Retrieved 2007-02-13.
- ↑ Stewart, Camille L. (2015). "The Fire at Cocoanut Grove:". Journal of Burn Care & Research 36 (1): 232–235. doi:10.1097/BCR.0000000000000111. https://academic.oup.com/jbcr/article/36/1/232-235/4568893.
- ↑ Ligon, B. Lee (2004). "Sir Alexander Fleming: Scottish researcher who discovered penicillin". Seminars in Pediatric Infectious Diseases 15 (1): 58–64. doi:10.1053/j.spid.2004.02.002. PMID 15175996. http://www.sciencedirect.com/science/article/pii/S1045187004000184.
- ↑ 104.0 104.1 Allison, V. D. (1974). "Personal recollections of Sir Almroth Wright and Sir Alexander Fleming". The Ulster Medical Journal 43 (2): 89–98. PMID 4612919. PMC 2385475. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2385475/.
- ↑ Bennett, Joan W; Chung, King-Thom (2001), "Alexander Fleming and the discovery of penicillin", Advances in Applied Microbiology, Elsevier, 49: 163–184, doi:10.1016/s0065-2164(01)49013-7, ISBN 978-0-12-002649-4, PMID 11757350, retrieved 2020-10-17
- ↑ Cairns, H.; Lewin, W. S.; Duthie, E. S.; Smith, HonorV. (1944). "Pneumococcal Meningitis Treated with Penicillin". The Lancet 243 (6299): 655–659. doi:10.1016/S0140-6736(00)77085-1. http://www.sciencedirect.com/science/article/pii/S0140673600770851.
- ↑ Fleming, Alexander (1943). "Streptococcal Meningitis treated With Penicillin.". The Lancet 242 (6267): 434–438. doi:10.1016/S0140-6736(00)87452-8. https://linkinghub.elsevier.com/retrieve/pii/S0140673600874528.
- ↑ Mathews, John A. (2008). "The Birth of the Biotechnology Era: Penicillin in Australia, 1943–80". Prometheus 26 (4): 317–333. doi:10.1080/08109020802459306. https://doi.org/10.1080/08109020802459306.
- ↑ Baldry, Peter (1976). The Battle Against Bacteria: A Fresh Look (in en). CUP Archive. p. 115. ISBN 978-0-521-21268-7. https://books.google.com/books?id=rvs8AAAAIAAJ.
- ↑ 110.0 110.1 Andrew Carroll (2014-06-02). "Here is Where: Penicillin Comes to Peoria". HistoryNet. Retrieved 2021-01-04.
- ↑ "Discovery and Development of Penicillin: International Historic Chemical Landmark". Washington, D.C.: American Chemical Society. Archived from the original on 28 June 2019. Retrieved 15 July 2019.
- ↑ 112.0 112.1 112.2 Neushul, P. (1993). "Science, government, and the mass production of penicillin". Journal of the History of Medicine and Allied Sciences 48 (4): 371–395. doi:10.1093/jhmas/48.4.371. PMID 8283024. https://pubmed.ncbi.nlm.nih.gov/8283024.
- ↑ Rodríguez-Sáiz, Marta; Díez, Bruno; Barredo, José Luis (2005). "Why did the Fleming strain fail in penicillin industry?". Fungal Genetics and Biology 42 (5): 464–470. doi:10.1016/j.fgb.2005.01.014. PMID 15809010. https://pubmed.ncbi.nlm.nih.gov/15809010.
- ↑ "Proteomics shows new faces for the old penicillin producer Penicillium chrysogenum". Journal of Biomedicine & Biotechnology 2012: 105109. 2012. doi:10.1155/2012/105109. PMID 22318718. PMC 3270403. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3270403/.
- ↑ Bentley, Ronald (2009). "Different roads to discovery; Prontosil (hence sulfa drugs) and penicillin (hence β-lactams)". Journal of Industrial Microbiology & Biotechnology 36 (6): 775–786. doi:10.1007/s10295-009-0553-8. PMID 19283418. http://link.springer.com/10.1007/s10295-009-0553-8.
- ↑ Kardos, Nelson; Demain, Arnold L. (2011). "Penicillin: the medicine with the greatest impact on therapeutic outcomes". Applied Microbiology and Biotechnology 92 (4): 677–687. doi:10.1007/s00253-011-3587-6. PMID 21964640. http://link.springer.com/10.1007/s00253-011-3587-6.
- ↑ Bauze, Robert (1997). "Editorial: Howard Florey and the penicillin story". Journal of Orthopaedic Surgery. Retrieved 2021-01-04.
- ↑ Taylor, Robert B. (2016), "Drugs and Other Remedies", White Coat Tales, Cham: Springer International Publishing, pp. 67–84, doi:10.1007/978-3-319-29055-3_3, ISBN 978-3-319-29053-9, retrieved 2021-01-04
- ↑ Raper, Kenneth B. (1946). "The development of improved penicillin‐producing molds". Annals of the New York Academy of Sciences 48 (2): 41–56. doi:10.1111/j.1749-6632.1946.tb31753.x. https://onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.1946.tb31753.x.
- ↑ Daemmrich, Arthur (2009). "Synthesis by microbes or chemists? Pharmaceutical research and manufacturing in the antibiotic era". History and Technology 25 (3): 237–256. doi:10.1080/07341510903083237. http://www.tandfonline.com/doi/abs/10.1080/07341510903083237.
- ↑ Wells, Percy A. (1991). "Penicillin Production Saga Recalled". Journal of the Washington Academy of Sciences 81 (3): 157–161. https://www.jstor.org/stable/24531093.
- ↑ Moyer, Andrew J.; Coghill, Robert D. (1946). "Penicillin". Journal of Bacteriology 51 (1): 79–93. doi:10.1128/jb.51.1.79-93.1946. PMID 16561059. PMC 518023. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC518023/.
- ↑ Wells, Percy A. (1975). "Some Aspects of the Early History of Penicillin in the United States". Journal of the Washington Academy of Sciences 65 (3): 96–101. https://www.jstor.org/stable/24536802.
- ↑ 124.0 124.1 Brown, W. E.; Peterson, W. H. (1950). "Factors Affecting Production of Penicillin in Semi-Pilot Plant Equipment". Industrial & Engineering Chemistry 42 (9): 1769–1774. doi:10.1021/ie50489a025. https://pubs.acs.org/doi/abs/10.1021/ie50489a025.
- ↑ Ligon, B. Lee (2004). "Penicillin: its discovery and early development". Seminars in Pediatric Infectious Diseases 15 (1): 52–57. doi:10.1053/j.spid.2004.02.001. PMID 15175995. https://pubmed.ncbi.nlm.nih.gov/15175995/.
- ↑ Savage, George M. (1949). "Improvement in streptomycin-producing strains of Streptomyces griseus by ultraviolet and X-ray energy". Journal of Bacteriology 57 (4): 429–441. doi:10.1128/jb.57.4.429-441.1949. PMID 16561716. PMC 385538. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC385538/.
- ↑ Raper, Kenneth B. (1952). "A Decade of Antibiotics in America". Mycologia 44 (1): 1–59. doi:10.1080/00275514.1952.12024170. https://www.tandfonline.com/doi/full/10.1080/00275514.1952.12024170.
- ↑ Stefaniak, J. J.; Gailey, F. B.; Brown, C. S.; Johnson, M. J. (1946). "Pilot Plant Equipment for Submerged Production of Penicillin". Industrial & Engineering Chemistry 38 (7): 666–671. doi:10.1021/ie50439a010. https://pubs.acs.org/doi/abs/10.1021/ie50439a010.
- ↑ Duckworth, R. B.; Harris, G. C. M. (1949). "The morphology of Penicillium chrysogenum in submerged fermentations". Transactions of the British Mycological Society 32 (3): 224–235. doi:10.1016/S0007-1536(49)80010-6. https://www.sciencedirect.com/science/article/pii/S0007153649800106.
- ↑ Goldschmidt, Millicent C.; Koffler, Henry (1950). "Effect of Surface-Active Agents on Penicillin Yields". Industrial & Engineering Chemistry 42 (9): 1819–1823. doi:10.1021/ie50489a035. https://pubs.acs.org/doi/abs/10.1021/ie50489a035.
- ↑ Hodgkin, D. C. (1949). "The X-ray analysis of the structure of penicillin". Advancement of Science 6 (22): 85–89. PMID 18134678. https://pubmed.ncbi.nlm.nih.gov/18134678.
- ↑ Crowfoot, D.; Bunn, C. W.; Rogers-Low, B. W.; Turner-Jones, A. (1949). "XI. The X-Ray Crystallographic Investigation of the Structure of Penicillin". In Clarke, Hans T.. Chemistry of Penicillin. Princeton University Press. pp. 310–366. doi:10.1515/9781400874910-012. ISBN 978-1-4008-7491-0. https://www.degruyter.com/document/doi/10.1515/9781400874910-012/html.
- ↑ Committee on Medical Research; Medical Research Council (1945). "Chemistry of penicillin". Science 102 (2660): 627–629. doi:10.1126/science.102.2660.627. PMID 17788243. https://pubmed.ncbi.nlm.nih.gov/17788243.
- ↑ Robinson, F. A. (1947). "Chemistry of penicillin". The Analyst 72 (856): 274. doi:10.1039/an9477200274. http://xlink.rsc.org/?DOI=an9477200274.
- ↑ 135.0 135.1 Wintersteiner, O.; Dutcher, J. D. (1949). "Chemistry of Antibiotics". Annual Review of Biochemistry 18 (1): 559–594. doi:10.1146/annurev.bi.18.070149.003015. http://www.annualreviews.org/doi/10.1146/annurev.bi.18.070149.003015.
- ↑ Carter, H. E.; Ford, J. H. (1950). "Biochemistry of antibiotics". Annual Review of Biochemistry 19 (1): 487–516. doi:10.1146/annurev.bi.19.070150.002415. http://www.annualreviews.org/doi/10.1146/annurev.bi.19.070150.002415.
- ↑ Ory, E. M. (1945). "Penicillin X: Comparison with penicillin g with respect to sensitivity of pathogenic organisms and serum levels". Journal of the American Medical Association 129 (4): 257. doi:10.1001/jama.1945.02860380015004. http://jama.jamanetwork.com/article.aspx?doi=10.1001/jama.1945.02860380015004.
- ↑ Schmidt, W. H.; Ward, G. E.; Coghill, R. D. (1945). "Penicillin: VI. Effect of dissociation phases of Bacillus subtilis on penicillin assay". Journal of Bacteriology 49 (4): 411–412. doi:10.1128/jb.49.4.411-412.1945. https://journals.asm.org/doi/10.1128/jb.49.4.411-412.1945.
- ↑ Chain, E (1948). "The chemistry of penicillin". Annual Review of Biochemistry 17 (1): 657–704. doi:10.1146/annurev.bi.17.070148.003301. PMID 18893607. http://www.annualreviews.org/doi/10.1146/annurev.bi.17.070148.003301.
- ↑ Roehr, M. (2000). "History of biotechnology in Austria". Advances in Biochemical Engineering/Biotechnology 69: 125–149. doi:10.1007/3-540-44964-7_5. PMID 11036693. https://pubmed.ncbi.nlm.nih.gov/11036693.
- ↑ Sheehan, John C.; H enery-Logan, Kenneth R. (March 5, 1957). "The Total Synthesis of Penicillin V". Journal of the American Chemical Society 79 (5): 1262–1263. doi:10.1021/ja01562a063.
- ↑ Sheehan, John C.; Henery-Loganm, Kenneth R. (June 20, 1959). "The Total Synthesis of Penicillin V". Journal of the American Chemical Society 81 (12): 3089–3094. doi:10.1021/ja01521a044.
- ↑ 143.0 143.1 Corey EJ, Roberts JD. "Biographical Memoirs: John Clark Sheehan". The National Academy Press. Retrieved January 28, 2013.
- ↑ Nicolaou, K. C.; Vourloumis, Dionisios; Winssinger, Nicolas; Baran, Phil S. (2000). "The Art and Science of Total Synthesis at the Dawn of the Twenty-First Century". Angewandte Chemie International Edition 39 (1): 44–122. doi:10.1002/(SICI)1521-3773(20000103)39:1<44::AID-ANIE44>3.0.CO;2-L. PMID 10649349. https://onlinelibrary.wiley.com/doi/abs/10.1002/%28SICI%291521-3773%2820000103%2939%3A1%3C44%3A%3AAID-ANIE44%3E3.0.CO%3B2-L.
- ↑ Sheehan, John C.; Logan, Kenneth R. Henery (1959). "A general synthesis of the penicillins". Journal of the American Chemical Society 81 (21): 5838–5839. doi:10.1021/ja01530a079.
- ↑ Sheehan, John C.; Henery-Logan, Kenneth R. (1962). "The Total and Partial General Syntheses of the Penicillins". Journal of the American Chemical Society 84 (15): 2983–2990. doi:10.1021/ja00874a029.
- ↑ Batchelor, F. R.; Doyle, F. P.; Nayler, J. H.; Rolinson, G. N. (1959). "Synthesis of penicillin: 6-aminopenicillanic acid in penicillin fermentations". Nature 183 (4656): 257–258. doi:10.1038/183257b0. PMID 13622762. https://pubmed.ncbi.nlm.nih.gov/13622762.
- ↑ Sheehan, J.C. (1964). "The Synthetic Penicillins". In Schueler, F.W.. Molecular Modification in Drug Design (in en). Advances in Chemistry. 45. Washington, D.C.: American Chemical Society. pp. 15–24. doi:10.1021/ba-1964-0045.ch002. ISBN 978-0-8412-0046-3.
- ↑ 149.0 149.1 Hamilton-Miller, J.M.T. (2008). "Development of the semi-synthetic penicillins and cephalosporins". International Journal of Antimicrobial Agents 31 (3): 189–92. doi:10.1016/j.ijantimicag.2007.11.010. PMID 18248798.
- ↑ Rolinson, G. N.; Geddes, A. M. (2007). "The 50th anniversary of the discovery of 6-aminopenicillanic acid (6-APA)". International Journal of Antimicrobial Agents 29 (1): 3–8. doi:10.1016/j.ijantimicag.2006.09.003. PMID 17137753. https://pubmed.ncbi.nlm.nih.gov/17137753.
- ↑ 151.0 151.1 Davies, J.; Davies, D. (2010). "Origins and evolution of antibiotic resistance". Microbiology and Molecular Biology Reviews 74 (3): 417–33. doi:10.1128/MMBR.00016-10. PMID 20805405. PMC 2937522. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2937522/.
- ↑ Harkins, Catriona P.; Pichon, Bruno; Doumith, Michel; Parkhill, Julian; Westh, Henrik; Tomasz, Alexander; de Lencastre, Herminia; Bentley, Stephen D. et al. (2017). "Methicillin-resistant Staphylococcus aureus emerged long before the introduction of methicillin into clinical practice". Genome Biology 18 (1): 130. doi:10.1186/s13059-017-1252-9. ISSN 1474-760X. PMID 28724393. PMC 5517843. https://pubmed.ncbi.nlm.nih.gov/28724393.
- ↑ Gaynes, Robert (2017). "The Discovery of Penicillin—New Insights After More Than 75 Years of Clinical Use". Emerging Infectious Diseases 23 (5): 849–853. doi:10.3201/eid2305.161556. PMC 5403050. http://wwwnc.cdc.gov/eid/article/23/5/16-1556_article.htm.
- ↑ 154.0 154.1 Abraham, Edward Penley (1983). "Ernst Boris Chain, 19 June 1906 - 12 August 1979". Biographical Memoirs of Fellows of the Royal Society 29: 42–91. doi:10.1098/rsbm.1983.0003. ISSN 0080-4606. https://royalsocietypublishing.org/doi/10.1098/rsbm.1983.0003.
- ↑ 155.0 155.1 Slinn, Judy (2008). "Patents and the UK pharmaceutical industry between 1945 and the 1970s". History and Technology 24 (2): 191–205. doi:10.1080/07341510701810963. ISSN 0734-1512. http://www.tandfonline.com/doi/abs/10.1080/07341510701810963.
- ↑ US 2442141, Template:Cite patent/authors, "Method for Production of Penicillin", issued 25 March 1948, assigned to US Agriculture
- ↑ US 2443989, Template:Cite patent/authors, "Method for Production of Penicillin", issued 22 June 1948, assigned to US Agriculture
- ↑ US 2476107, Template:Cite patent/authors, "Method for Production of Penicillin", issued 12 July 1949, assigned to US Agriculture
- ↑ Tyabji, Nasir (2004). "Gaining Technical Know-How in an Unequal World: Penicillin Manufacture in Nehru's India". Technology and Culture 45 (2): 331–349. doi:10.1353/tech.2004.0097. https://mpra.ub.uni-muenchen.de/84236/1/MPRA_paper_84236.pdf.
- ↑ Allison, V. D. (1974). "Personal recollections of Sir Almroth Wright and Sir Alexander Fleming.". The Ulster Medical Journal 43 (2): 89–98. PMID 4612919. PMC 2385475. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2385475/.
- ↑ "The Nobel Prize in Physiology or Medicine 1945". NobelPrize.org. Retrieved 2020-07-26.
- ↑ "The Nobel Prize in Chemistry 1964". NobelPrize.org. Retrieved 2021-10-11.
- ↑ Kirby, W. M.; Bulger, R. J. (1964). "The new penicillins and cephalosporins". Annual Review of Medicine 15: 393–412. doi:10.1146/annurev.me.15.020164.002141. PMID 14133855. https://pubmed.ncbi.nlm.nih.gov/14133855.
- ↑ 164.0 164.1 Acred, P.; Brown, P.; Turner, D.H.; Wilson, M.J. (1962). "Pharmacology and chemotherapy of ampicillin--a new broad-spectrum penicillin". British Journal of Pharmacology and Chemotherapy 18 (2): 356–69. doi:10.1111/j.1476-5381.1962.tb01416.x. PMID 13859205. PMC 1482127. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1482127/.
- ↑ "Methicillin-Resistant Staphylococci in a General Hospital". Lancet 1 (7385): 595–7. March 1965. doi:10.1016/S0140-6736(65)91165-7. PMID 14250094.
- ↑ de Sousa Coelho, F.; Mainardi, J.-L. (2021-01-05). "The multiple benefits of second-generation β-lactamase inhibitors in treatment of multidrug-resistant bacteria". Infectious Diseases Now online. doi:10.1016/j.idnow.2020.11.007. PMID 33870896. https://pubmed.ncbi.nlm.nih.gov/33870896.
- ↑ Croydon, E. A.; Sutherland, R. (1970). "α-amino-p-hydroxybenzylpenicillin (BRL 2333), a new semisynthetic penicillin: absorption and excretion in man". Antimicrobial Agents and Chemotherapy 10: 427–430. PMID 5521362. https://pubmed.ncbi.nlm.nih.gov/5521362.
- ↑ Sutherland, R.; Rolinson, G. N. (1970). "α-amino-p-hydroxybenzylpenicillin (BRL 2333), a new semisynthetic penicillin: in vitro evaluation". Antimicrobial Agents and Chemotherapy 10: 411–415. doi:10.1128/AAC.10.3.411. PMID 5000265. https://pubmed.ncbi.nlm.nih.gov/5000265.
- ↑ Burch, D. G. S.; Sperling, D. (2018). "Amoxicillin-current use in swine medicine". Journal of Veterinary Pharmacology and Therapeutics 41 (3): 356–368. doi:10.1111/jvp.12482. http://doi.wiley.com/10.1111/jvp.12482.
- ↑ Aberer, Werner; Macy, Eric (2017). "Moving toward optimizing testing for penicillin allergy". The Journal of Allergy and Clinical Immunology. In Practice 5 (3): 684–685. doi:10.1016/j.jaip.2017.03.020. PMID 28483319. https://pubmed.ncbi.nlm.nih.gov/28483319.
- ↑ James, C.W.; Gurk-Turner, C. (2001). "Cross-reactivity of beta-lactam antibiotics". Proceedings 14 (1): 106–7. doi:10.1080/08998280.2001.11927741. PMID 16369597. PMC 1291320. //www.ncbi.nlm.nih.gov/pmc/articles/PMC1291320/.
- ↑ Lima, Lidia Moreira; Silva, Bianca Nascimento Monteiro da; Barbosa, Gisele; Barreiro, Eliezer J. (2020). "β-lactam antibiotics: An overview from a medicinal chemistry perspective". European Journal of Medicinal Chemistry 208: 112829. doi:10.1016/j.ejmech.2020.112829. PMID 33002736. https://pubmed.ncbi.nlm.nih.gov/33002736.
- ↑ Abraham, E.P.; Chain, E. (1940). "An enzyme from bacteria able to destroy penicillin". Nature 10 (4): 677–8. doi:10.1038/146837a0. PMID 3055168. http://www.nature.com/articles/146837a0.
- ↑ Lowy, F.D. (2003). "Antimicrobial resistance: the example of Staphylococcus aureus". The Journal of Clinical Investigation 111 (9): 1265–73. doi:10.1172/JCI18535. PMID 12727914. PMC 154455. //www.ncbi.nlm.nih.gov/pmc/articles/PMC154455/.
- ↑ Davies, J; Davies, D (2010). "Origins and evolution of antibiotic resistance". Microbiology and Molecular Biology Reviews 74 (3): 417–33. doi:10.1128/MMBR.00016-10. PMID 20805405. PMC 2937522. //www.ncbi.nlm.nih.gov/pmc/articles/PMC2937522/.

