WikiJournal of Science/The TIM barrel fold
WikiJournal of Science
Open access • Publication charge free • Public peer review • Wikipedia-integrated
Previous
Volume 1(1)
Volume 1(2)
Volume 2(1)
Volume 3(1)
Volume 4(1)
Volume 5(1)
Volume 6(1)
This article has been through public peer review.
Post-publication review comments or direct edits can be left at the version as it appears on Wikipedia.First submitted:
Accepted:
Reviewer comments
PDF: Download
DOI: 10.15347/wjs/2020.004
QID: Q87400003
XML: Download
Share article
![]() Email
|
Email
| ![]() Facebook
|
Facebook
| ![]() Twitter
|
Twitter
| ![]() LinkedIn
|
LinkedIn
| ![]() Mendeley
|
Mendeley
| ![]() ResearchGate
ResearchGate
Suggested citation format:
Deepesh Nagarajan; Neha Nanajkar (2020). "The TIM barrel fold". WikiJournal of Science 3 (1): 4. doi:10.15347/WJS/2020.004. Wikidata Q87400003. ISSN 2470-6345. https://upload.wikimedia.org/wikiversity/en/c/c1/The_TIM_barrel_fold.pdf.
Citation metrics
AltMetrics
Page views on Wikipedia
Wikipedia: Content from this work is used in the following Wikipedia article: TIM barrel.
License: ![]()
![]() This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
Hemachander Subramanian ![]() (handling editor) contact
(handling editor) contact
Thomas Shafee ![]() contact
contact
Reinhard Sterner ![]()
C. Robert Matthews
Cristina Elisa Martina
Article information
Abstract
Introduction
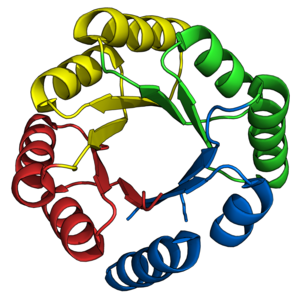
The TIM barrel is a structurally conserved protein fold, named after triose phosphate isomerase, a constituent enzyme of the glycolysis pathway whose structure was first solved in 1975.[1] TIM barrels contain 200-250 amino acid residues,[2] which form 8 β-strands and 8 α-helices. The β-strands are arranged into a parallel β-barrel, and are surrounded by the 8 α-helices. The inner β-barrel is in many cases stabilized by intricate salt-bridge networks.[3] Loops at the C-terminal ends of the β-barrel are responsible for catalytic activity[4][5] while N-terminal loops are important for the stability of the TIM-barrels. Structural inserts ranging from extended loops to independent domains may be inserted in place of these loops or at the N/C-terminals. TIM barrels are ubiquitous, with approximately 10% of all enzymes adopting this fold.[6] Further, 5 of 7 enzyme commission (EC) enzyme classes include TIM barrel proteins.[7][8] The TIM barrel fold is evolutionarily ancient, with many of its members possessing little similarity today,[9] instead falling within the twilight zone of sequence similarity.[10][11] TIM barrels appear to have evolved through gene duplication and domain fusion events of half-barrel proteins,[12] with a majority of TIM barrels originating from a common ancestor. This lead many TIM barrels to possess internal symmetries.[13] Further gene duplication events of this ancestral TIM barrel lead to diverging enzymes possessing the functional diversity observed today. TIM barrels have also been a longstanding target for protein designers. Successful TIM barrel designs include both domain fusions of existing proteins and de novo designs. Domain fusions experiments have resulted in many successful designs,[14][15][16][17][18][19][20] whereas de novo designs only yielded successes after 28 years of incremental development.[21]
Structure
and blue. Each layer contains 4 residues that point towards the pore, and lie on the same plane perpendicular to the barrel axis. The shear number for TIM barrels is always 8, and is illustrated in magenta. Some TIM barrels naturally adopt, or are designed to adopt, two or four-fold symmetry. Example asymmetric units are also highlighted. This figure has been adapted with permission from previously published work.[22]
Topology
The TIM barrel gets its name from the enzyme triose phosphate isomerase (TIM), which was the first protein possessing the fold to be crystallized.[1] TIM barrels (Figure 1A) contain 200-250 amino acid residues,[2] folded into 8 α-helices and 8 β-strands. The β-strands are arranged into a parallel β-barrel. The defining property of TIM β-barrels is that they always possess a shear number of 8.[2] The shear number is determined by picking a residue x on β-strand-1, and moving along the β-barrel, in a perpendicular direction to the direction of the strands, until residue y on the original β-strand-1 is reached. The number of residues between the start and end positions (|y−x|) is the shear number.[23] Since the number of strands is equal to the Shear number, side-chains point alternatively towards the pore and the core, giving a 4-fold symmetry. The α-helices surround and completely enclose the inner β-barrel. Short loops typically connect the α and β secondary structures, forming a (βα)8 repeat topology. In some cases, structures ranging from extended loops to independent domains may be inserted in place of these loops, or may be attached to the N/C-terminals. All TIM barrel enzymes possess catalytic sites at the C-terminal end of the β-barrel,[24] and structural inserts present close to this end may aid in catalytic activity. The overall topology of the TIM barrel is illustrated in Figure 2.
Core and pore regions
TIM barrels contain two distinct buried regions, where amino acid residues are completely enveloped by their neighbors and lack access to solvent. We have previously referred to these regions as the 'core' and 'pore',[22] and are depicted in Figure 1B. It should be noted that the term 'pore' is a misnomer, as no solvent channels exist within this region. The core region consists of all residues constituting the α-β interface, and lies exterior to the central β-barrel. The pore region consists of all interior β-barrel residues, which are surrounded and enclosed by the β-barrel backbone.
Due to the pleated nature of β-strands, alternate residues along a strand are almost evenly split between the pore (53%) and core (47%).[22] For β-barrels, 95% of their core residues are buried. Only 11% of their core residues are polar, possessing an affinity for water, and possessing the ability to form hydrogen bonds or salt bridges. Similarly, 84% of β-strand pore residues are buried. However, 42% of their pore residues are polar. These residues form intricate salt bridge networks to compensate for their lack of solvent accessibility.
TIM barrel stabilizing elements
Salt bridges within TIM barrel pores are thought to contribute to the overall stability of the fold. An example of a large salt bridge network can be found in 2-deoxyribose-5-phosphate aldolase (Figure 3). This network was found to be conserved across the Class I aldolase family.[3]
The exact reason for the overrepresentation of polar residues and salt bridges within the pore remains unclear. One study proposes that they improve foldability rather than thermodynamic stability of TIM barrels.[22] During the folding process, inner pore residues on β-strands would be exposed to water. Partially-folded βαβα modules, called foldons, would be energetically stabilized by polar pore residues during this stage of folding.
In another study involving the S. solfataricus indole-3-glycerol phosphate synthase TIM barrel protein, a conserved βαβαβ module was found to be an essential folding template, which guided the folding of other secondary structures.[25] β-barrel closure only occurred at the end of the folding process. In this case however, the authors credited branched aliphatic amino acids (valine, leucine, and isoleucine) for foldon stability.
Another stabilizing element in TIM barrels is the β-hairpin clamp. Side chain H-bond donors at the N-termini of even-numbered β-strands often form H-bonds with main chain amide hydrogens in preceding odd-numbered β-strands.[26][27] These clamps (or hydrophobic side chain bridge analogs) are conserved in 3 indole-3-glycerolphosphate synthase TIM barrel orthologs from the bacterial and archaeal kingdoms,[28] implying they arose in their last common ancestor and have been preserved for over a billion years.
Structural inserts
The N/C-terminal and loop regions on TIM barrel proteins are capable of hosting structural inserts ranging from simple secondary structural motifs to complete domains. These domains aid in substrate recognition and catalytic activity. Four diverse examples of TIM barrels containing additional motifs and domains are discussed below.
Bacillus subtilis Orotidine 5'-phosphate decarboxylase[30] (PDB: 1DBT, Figure 4A) is a TIM barrel protein displaying 4 α-helices in place of the βα loops typically present at the C-terminal of the β-barrel (residues 35-42, 89-91, 126-133, and 215-219). One of these helices (R215→K219) contains a conserved arginine residue (R215) required for interacting with a phosphate moiety on orotidine 5′-monophosphate. The other helices were not found to host residues critical for catalytic activity, and may serve in structural roles.
Mycobacterium tuberculosis bifunctional histidine/tryptophan biosynthesis isomerase (PriA) (PDB: 2Y85, Figure 4B) possesses the ability to catalyse two reactions: (i) HisA reaction: the conversion of N-[(5-phosphoribosyl) formimino]-5-aminoimidazole-4-carboxamide ribonucleotide (ProFAR) to N-[(5-phosphoribulosyl)formimino]-5-aminoimidazole-4-carboxamide ribonucleotide (PRFAR), and (ii) TrpF reaction: N-(5’-phosphoribosyl)-anthranilate (PRA) to 1-(O-carboxyphenylamino)- 1’-deoxyribulose-5’-phosphate (CdRP).[31] PriA is a TIM barrel enzyme that accommodates both substrates using active site loops (loops 1, 5, and 6, extended βα loops at the C-terminal end of the β-barrel) that change conformation depending on the reactant present.[32] Loop 1 wraps over the active site only in the presence of ProFAR. Loop5 wraps over the active site, adopting a β-sheet conformation in the presence of CdRP, or a knot-like conformation in the presence of ProFAR. Loop 6 wraps over the active site for all reactants.
Lactococcus lactis Dihydroorotate dehydrogenase A (DHODA) (PDB: 2DOR, Figure 4C) is an example of a TIM barrel possessing β-sheets and extended loops over the C-terminal end of the β-barrel. DHODA catalyzes the oxidation of dihydroorotate to orotate,[33][34] which is part of the de novo uridine 5'-monophosphate (UMP) synthesis pathway. This oxidation is mediated by flavin mononucleotide (FMN). Here, β-sheets and extended loops enclose the active site forming a cavity, while also hosting several catalytic residues.
The Methylophilus methylotrophus trimethylamine dehydrogenase[29] (PDB: 2TMD, Figure 4D) TIM barrel is an example of a complete domain insertion. Here, a Rossmann fold domain is inserted at the C-terminal end of the TIM-barrel. Trimethylamine dehydrogenase catalyzes the conversion of trimethylamine to formaldehyde.[35] This reaction requires both a reduced 6-S-cysteinyl Flavin mononucleotide (FMN) cofactor and a reduced iron-sulphur ([4Fe-4S]+) center. FMN is covalently bound within the C-terminal region of the β-barrel. The [4Fe-4S]+ center is too large to be accommodated within the TIM barrel, and is instead placed in close proximity, 7 Å away, at the interface between the TIM barrel and Rossmann fold domains.
Folding mechanisms
The conservation of the TIM barrel fold is mirrored by the conservation of its equilibrium and kinetic folding mechanisms in bacterial paralogs with phylogenetically distinct lineages. Chemical denaturation of several natural[36][37] and 2 designed TIM barrel variants[37] invariably involves a highly populated equilibrium intermediate. The kinetic intermediates that appear after dilution from highly denaturing solutions involve an early misfolded species that must at least partially unfold to access the productive folding pathway.[36][37] The rate-limiting step in folding is the closure of the 8-stranded β-barrel, with the preceding, open barrel form corresponding to the equilibrium intermediate(Figure 5).[38] Native-centric molecular dynamics simulations recapitulate the experimental results and point the way to testable computational models for complex folding mechanisms.[39]
Conserved fitness landscapes
TIM barrel proteins possess an unusually high sequence plasticity, forming large families of orthologous and paralogous enzymes in widely divergent organisms. This plasticity suggests a sequence landscape that allows for protein adaptation to a variety of environmental conditions, largely independent of phylogenetic history, while maintaining function. Chan et al.[28] used a deep mutational scanning approach and a competition assay[40] to determine the fitness of all possible amino acid mutants across positions in 3 hyperthermophilic indole-3-glycerolphosphate synthase (IGPS) TIM barrel enzymes in supporting the growth of a yeast host lacking IGPS. Although the 2 bacterial and 1 archaeal IGPS enzymes were only 30-40% identical in sequence, their fitness landscapes were strongly correlated: the same amino acids at the same positions in the three different proteins had very similar fitness. The correlation can be thought of as the conservation of the fitness landscape for a TIM barrel enzyme across evolutionary time (Figure 6).
Evolution and origins
The predominant theory for TIM barrel evolution involves gene duplication and fusion, starting with a half- barrel that eventually formed a full TIM barrel. Multiple studies support the theory of divergent evolution from a single ancestor, and are discussed below.
Evolution from a common ancestor
In the early 1990s, Farber et al.[4][5] noted that all TIM barrel structures solved at the time were enzymes, indicating divergence from a common ancestor. Further, all TIM barrels possessed active sites at the C-terminal end of β-barrels. Brändén[24] suggested that a common phosphate binding site, formed by a small α-helix and TIM barrel loops-7/8, strongly indicated divergent evolution. Copley et al.[41] further studied these phosphate groups, concluding that 12 of 23 SCOP (structural classification of proteins)[42] TIM barrel families diverged from a common ancestor. Similarly, Nagano et al.[9] concluded that there were hints for common ancestry for 17 of the 21 CATH (classification of protein domain structures)[43] TIM barrel families. Based on these reports, it is plausible that the majority of TIM barrel proteins evolved from a common ancestor.
Origin through gene duplication and domain fusion
Many TIM barrel proteins possess 2-fold, 4-fold or 8-fold internal symmetry, suggesting that TIM barrels evolved from ancestral (βα)4, (βα)2, or βα motifs through gene duplication and domain fusion. A good example of 2-fold internal symmetry is observed in the enzymes ProFAR isomerase (HisA) and imidazole glycerol phosphate synthase (HisF) of the Thermotoga maritima histidine biosynthesis pathway.[12] They catalyze 2 successive reactions in the pathway, possess 25% sequence homology, and possess root-mean-square deviations (RMSDs) between 1.5-2 Å, suggesting divergence from a common ancestor. More interestingly, the loops on the C terminal ends of both HisA and HisF showed a twofold repeated pattern, suggesting that their common ancestor also possessed 2-fold internal symmetry. Using these observations, Lang et al.[12] constructed a model for the evolution of the TIM barrels (Figure 5). An ancestral half-barrel would have undergone a gene duplication and fusion event, resulting in a single protein containing two half-barrel domains. Structural adaptations would have occurred, resulting in the merging of these domains to form a closed β-barrel, and forming an ancestral TIM barrel. Functional adaptations would have also occurred, resulting in the evolution of new catalytic activity at the C terminal end of the β-barrel. At this point, the common ancestor of HisA and HisF would have undergone a second gene duplication event. Divergent evolution of the duplicated genes of the ancestral TIM barrel would have resulted in the formation of HisA and HisF.
Interestingly, this evolutionary model has been experimentally validated using rational protein design and directed evolution. Höcker et al. first fused two C-terminal halves of HisF, yielding HisF-CC. This construct was then stabilized by the insertion of an internal salt-bridge, yielding HisF-C*C.[16] Seitz et al.[14] and Höcker et al.[15] then stepwise further stabilized and solubilized HisF-C*C by optimizing the half-barrel interface, generating HisF-C**C and HisF-C***C, respectively. The crystal structure of HisF-C***C revealed a 2-fold symmetric TIM barrel, validating the possibility of natural domain fusion. Moreover, Höcker created the first chimeric HisAF and HisFA TIM barrels using HisA and HisF half-barrels.[16] These experiments led to the proposal of a novel means of diversification and evolution of TIM-barrel enzymes through the exchange of (βα)4 half-barrel domains amongst preexisting TIM barrels. In accordance with this idea, Claren et al. established high catalytic activity on the HisAF construct.[17] Similarly, chimeric βα5-flavodoxin-like fold (CheY)/HisF TIM barrels,[18][19] and a perfectly 2-fold symmetric HisF-based TIM barrel[20][37] have also been created.
The existence of 4/8-fold internal symmetry was suggested by Soding et al.[13] based on a computational analysis of TIM barrel sequences. For example, Escherichia coli KDPG aldolase[44] (PDB: 1FQ0) was suggested to possess a distinct 4-fold symmetry, with discernible 8-fold symmetry. The design of a 4-fold symmetric TIM barrel[21] confirmed the possibility of higher orders of internal symmetry in natural TIM barrels, and will be discussed in detail in the next section. It should be noted that no experimental evidence for the existence of 8-fold symmetric TIM barrels has been reported to date.
De novo TIM barrel design
The TIM barrel fold has been a long-standing target for de novo protein designers. As previously described, numerous TIM barrels have been successfully designed based on preexisting natural half-barrels. In contrast, the de novo design of TIM barrels occurred in incremental steps over a period of 28 years.[45]
The Octarellin series[46][47][48][49][50] of proteins (Octarellin I→VI) were the first attempts to create a de novo TIM barrel. As the field of protein design was still in its infancy, these design attempts were only met with limited success. Although they displayed circular dichroism spectra consistent with αβ proteins and some cooperative folding characteristics, all Octarellin series peptides were insoluble, and had to be resolubilized from inclusion bodies for further characterization. Interestingly, Octarellin V.1[51] displayed a Rossmann-like fold under co-crystal conditions.
The Symmetrin series[22] of proteins (Symmetrin-1→4) displayed more favorable biophysical characteristics. Symmetrin-1 was readily soluble, displayed circular dichroism spectra consistent with αβ proteins, and displayed excellent cooperative unfolding and refolding characteristics. Despite these advances, all proteins in this family displayed molten characteristics when analyzed using NMR (nuclear magnetic resonance), and further work to solve their structures could not be pursued.
Proteins of the sTIM series[21] represented the first successful de novo TIM barrel design.[52][45] sTIM-11 (PDB: 5BVL) was designed with an internal 4-fold symmetry, to reduce the complexity of computational design using the Rosetta software suite.[53] Previously-derived first principles[54] were used to delineate secondary structure topologies and lengths. sTIM-11 proved to be a highly thermostable, cooperatively folding design that adopted its intended structure (Figure 6).
Additional information
Acknowledgements
The authors thank our open reviewer Robert Matthews, with the assistance of Yvonne Chan and Konstantin Zeldovich, for contributing additional sections for this review (folding mechanisms, conserved fitness landscapes, βα stabilizing clamp).
References
- ↑ 2.0 2.1 2.2 Wierenga, RK (2001). "The TIM-barrel fold: a versatile framework for efficient enzymes". FEBS Letters 492 (3): 193-198. doi:10.1016/s0014-5793(01)02236-0.
- ↑ 3.0 3.1 Vijayabaskar, MS; Vishveshwara, Saraswathi (2012). "Insights into the fold organization of TIM barrel from interaction energy based structure networks". PLoS Computational Biology 8 (5): e1002505. doi:10.1371/journal.pcbi.1002505.
- ↑ 4.0 4.1 Farber, Gregory K; Petsko, Gregory A (1990). "The evolution of α/β barrel enzymes". Trends in Biochemical Sciences 15 (6): 228-234. doi:10.1016/0968-0004(90)90035-A.
- ↑ 5.0 5.1 Reardon, D; Farber, GK (1995). "The structure and evolution of alpha/beta barrel proteins". The FASEB Journal 9 (7): 497-503. doi:10.1096/fasebj.9.7.7737457.
- ↑ Jansen, Ronald; Gerstein, Mark (2000). "Analysis of the yeast transcriptome with structural and functional categories: characterizing highly expressed proteins". Nucleic Acids Research 28 (6): 1481-1488. doi:10.1093/nar/28.6.1481.
- ↑ Nagano, Nozomi; Hutchinson, E Gail; Thornton, Janet M (1999). "Barrel structures in proteins: automatic identification and classification including a sequence analysis of TIM barrels". Protein Science 8 (10): 2072-2084. doi:10.1110/ps.8.10.2072.
- ↑ Webb, Edwin C. (1992). Enzyme nomenclature: Recommendations of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology on the Nomenclature and Classification of Enzymes. Academic Press. ISBN 978-0-12-227164-9.
- ↑ 9.0 9.1 Nagano, Nozomi; Orengo, Christine A; Thornton, Janet M (2002). "One fold with many functions: the evolutionary relationships between TIM barrel families based on their sequences structures and functions". Journal of molecular biology 321 (5): 741-765. doi:10.1016/s0022-2836(02)00649-6.
- ↑ Livesay, Dennis R; La, David (2005). "The evolutionary origins and catalytic importance of conserved electrostatic networks within TIM-barrel proteins". Protein Science 14 (5): 1158-1170. doi:10.1110/ps.041221105.
- ↑ Chung, Su Yun; Subbiah, S (1996). "A structural explanation for the twilight zone of protein sequence homology". Structure 4 (10): 1123-1127. doi:10.1016/s0969-2126(96)00119-0.
- ↑ 12.0 12.1 12.2 12.3 Lang, Dietmar; Thoma, Ralf; Henn-Sax, Martina; Sterner, Reinhard; Wilmanns, Matthias (2000). "Structural evidence for evolution of the β/α barrel scaffold by gene duplication and fusion". Science 289 (5484): 1546-1550. doi:10.1126/science.289.5484.1546.
- ↑ 13.0 13.1 Soding, Johannes; Remmert, Michael; Biegert, Andreas (2006). "HHrep: de novo protein repeat detection and the origin of TIM barrels". Nucleic Acids Research 34 (Suppl 2): W137-W142. doi:10.1093/nar/gkl130.
- ↑ 14.0 14.1 Seitz, Tobias; Bocola, Marco; Claren, Jorg; Sterner, Reinhard (2007). "Stabilisation of a (βα)8-barrel protein designed from identical half barrels". Journal of Molecular Biology 372 (1): 114-129. doi:10.1016/j.jmb.2007.06.036.
- ↑ 15.0 15.1 Hocker, Birte; Lochner, Adriane; Seitz, Tobias; Claren, Jorg; Sterner, Reinhard (2009). "High-resolution crystal structure of an artificial (βα)8-barrel protein designed from identical half-barrels". Biochemistry 48 (6): 1145-1147. doi:10.1021/bi802125b.
- ↑ 16.0 16.1 16.2 Hocker, Birte; Claren, Jorg; Sterner, Reinhard (2004). "Mimicking enzyme evolution by generating new (βα) 8-barrels from (βα) 4-half-barrels". Proceedings of the National Academy of Sciences 101 (47): 16448-16453. doi:10.1073/pnas.0405832101.
- ↑ 17.0 17.1 Claren, Jorg; Malisi, Christoph; Hocker, Birte; Sterner, Reinhard (2009). "Establishing wild-type levels of catalytic activity on natural and artificial (βα)8-barrel protein scaffolds". Proceedings of the National Academy of Sciences 106 (10): 3704-3709. doi:10.1073/pnas.0810342106.
- ↑ 18.0 18.1 Bharat, Tanmay AM; Eisenbeis, Simone; Zeth, Kornelius; Hocker, Birte (2008). "A βα-barrel built by the combination of fragments from different folds". Proceedings of the National Academy of Sciences 105 (29): 9942-9947. doi:10.1073/pnas.0802202105.
- ↑ 19.0 19.1 Eisenbeis, Simone; Proffitt, William; Coles, Murray; Truffault, Vincent; Shanmugaratnam, Sooruban; Meiler, Jens; Hocker, Birte (2012). "Potential of fragment recombination for rational design of proteins". Journal of the American Chemical Society 134 (9): 4019-4022. doi:10.1021/ja211657k.
- ↑ 20.0 20.1 Fortenberry, Carie; Bowman, Elizabeth Anne; Proffitt, Will; Dorr, Brent; Combs, Steven; Harp, Joel; Mizoue, Laura; Meiler, Jens (2011). "Exploring symmetry as an avenue to the computational design of large protein domains". Journal of the American Chemical Society 133 (45): 18026-18029. doi:10.1021/ja210593m.
- ↑ 21.0 21.1 21.2 Huang, Po-Ssu; Feldmeier, Kaspar; Parmeggiani, Fabio; Velasco, D Alejandro Fernandez; Hcker, Birte; Baker, David (2016). "De novo design of a four-fold symmetric TIM-barrel protein with atomic-level accuracy". Nature Chemical Biology 12 (1): 29. doi:10.1038/nchembio.1966.
- ↑ 22.0 22.1 22.2 22.3 22.4 Nagarajan, Deepesh; Deka, Geeta; Rao, Megha (2015). "Design of symmetric TIM barrel proteins from first principles". BMC Biochemistry 16 (1): 18. doi:10.1186/s12858-015-0047-4.
- ↑ Murzin, Alexey G; Lesk, Arthur M; Chothia, Cyrus (1994). "Principles determining the structure of β-sheet barrels in proteins I. A theoretical analysis". Journal of Molecular Biology 236 (5): 1369-1381. doi:10.1016/0022-2836(94)90064-7.
- ↑ 24.0 24.1 Brändén, Carl-Ivar (1991). "The TIM barrel—the most frequently occurring folding motif in proteins". Current Opinion in Structural Biology 1 (6): 978-983. doi:10.1016/0959-440x(91)90094-a.
- ↑ Gu, Zhenyu; Zitzewitz, Jill A; Matthews, C Robert (2007). "Mapping the structure of folding cores in TIM barrel proteins by hydrogen exchange mass spectrometry: the roles of motif and sequence for the indole-3-glycerol phosphate synthase from Sulfolobus solfataricus". Journal of Molecular Biology 368 (2): 582-594. doi:10.1016/j.jmb.2007.02.027.
- ↑ Yang, Xiaoyan; Vadrevu, Ramakrishna; Wu, Ying; Matthews, CRobert (2007). "Long-range side-chain--main-chain interactions play crucial roles in stabilizing the (βα) 8 barrel motif of the alpha subunit of tryptophan synthase". Protein Science 16 (7): 1398-1409. doi:10.1110/ps.062704507.
- ↑ Yang, Xiaoyan; Kathuria, Sagar V; Vadrevu, Ramakrishna; Matthews, C Robert (2009). "βα-Hairpin clamps brace βαβ modules and can make substantive contributions to the stability of TIM barrel proteins". PLoS One 4 (9). doi:10.1371/journal.pone.0007179.
- ↑ 28.0 28.1 Chan, Yvonne H; Venev, Sergey V; Zeldovich, Konstantin B; Matthews, C Robert (2017). "Correlation of fitness landscapes from three orthologous TIM barrels originates from sequence and structure constraints". Nature Communications 8 (1): 1-12. doi:10.1038/ncomms14614.
- ↑ 29.0 29.1 Barber, Michael J; Neame, Peter J; Lim, Louis W; White, Scott; Matthews, FS (1992). "Correlation of x-ray deduced and experimental amino acid sequences of trimethylamine dehydrogenase". Journal of Biological Chemistry 267 (10): 6611-6619. doi:10.1016/S0021-9258(19)50471-9.
- ↑ Appleby, Todd C; Kinsland, Cynthia; Begley, Tadhg P; Ealick, Steven E (2000). "The crystal structure and mechanism of orotidine 5'-monophosphate decarboxylase". Proceedings of the National Academy of Sciences 97 (5): 2005-2010. doi:10.1073/pnas.259441296.
- ↑ Juarez-Vazquez, Ana Lilia; Edirisinghe, Janaka N; Verduzco-Castro, Ernesto A; Michalska, Karolina; Wu, Chenggang; Noda-Garcia, Lianet; Babnigg, Gyorgy; Endres, Michael et al. (2017). "Evolution of substrate specificity in a retained enzyme driven by gene loss". eLife 6: e22679. doi:10.7554/elife.22679.
- ↑ Due, Anne V; Kuper, Jochen; Geerlof, Arie; von Kries, Jens Peter; Wilmanns, Matthias (2011). "Bisubstrate specificity in histidine/tryptophan biosynthesis isomerase from Mycobacterium tuberculosis by active site metamorphosis". Proceedings of the National Academy of Sciences 108 (9): 3554-3559. doi:10.1073/pnas.1015996108.
- ↑ Rowland, Paul; Larsen, Sine; Bjornberg, Olof; Nielsen, Finn S; Jensen, Kaj Frank (1998). "The crystal structure of Lactococcus lactis dihydroorotate dehydrogenase A complexed with the enzyme reaction product throws light on its enzymatic function". Protein Science 7 (6): 1269-1279. doi:10.1002/pro.5560070601.
- ↑ Bjornberg, Olof; Rowland, Paul; Larsen, Sine; Jensen, Kaj Frank (1997). "Active site of dihydroorotate dehydrogenase A from Lactococcus lactis investigated by chemical modification and mutagenesis". Biochemistry 36 (51): 16197-16205. doi:10.1021/bi971628y.
- ↑ Jang, Mei-Huei; Basran, Jaswir; Scrutton, Nigel S; Hille, Russ (1999). "The reaction of trimethylamine dehydrogenase with trimethylamine". Journal of Biological Chemistry 274 (19): 13147-13154. doi:10.1074/jbc.274.19.13147.
- ↑ 36.0 36.1 Forsyth, William R; Bilsel, Osman; Gu, Zhenyu; Matthews, C Robert (2007). "Topology and sequence in the folding of a TIM barrel protein: global analysis highlights partitioning between transient off-pathway and stable on-pathway folding intermediates in the complex folding mechanism of a (βα) 8 barrel of unknown function from B. subtilis". Journal of Molecular Biology 372 (1): 236-253. doi:10.1016/j.jmb.2007.06.018.
- ↑ 37.0 37.1 37.2 37.3 Carstensen, Linn; Sperl, Josef M; Bocola, Marco; List, Felix; Schmid, Franz X; Sterner, Reinhard (2012). "Conservation of the folding mechanism between designed primordial (βα) 8-barrel proteins and their modern descendant". Journal of the American Chemical Society 134 (30): 12786-12791. doi:10.1021/ja304951v.
- ↑ Gu, Zhenyu; Rao, Maithreyi K; Forsyth, William R; Finke, John M; Matthews, C Robert (2007). "Structural analysis of kinetic folding intermediates for a TIM barrel protein, indole-3-glycerol phosphate synthase, by hydrogen exchange mass spectrometry and G{\=o". Journal of Molecular Biology 374 (2): 528-546. doi:10.1016/j.jmb.2007.09.024.
- ↑ Halloran, Kevin T; Wang, Yanming; Arora, Karunesh; Chakravarthy, Srinivas; Irving, Thomas C; Bilsel, Osman; Brooks, Charles L; Matthews, C Robert (2019). "Frustration and folding of a TIM barrel protein". Proceedings of the National Academy of Sciences 116 (33): 16378-16383. doi:10.1073/pnas.1900880116.
- ↑ Hietpas, Ryan T; Jensen, Jeffrey D; Bolon, Daniel NA (2011). "Experimental illumination of a fitness landscape". Proceedings of the National Academy of Sciences 108 (19): 7896-7901. doi:10.1073/pnas.1016024108.
- ↑ Copley, Richard R; Bork, Peer (2000). "Homology among (βα) 8 barrels: implications for the evolution of metabolic pathways". Journal of Molecular Biology 303 (4): 627-641. doi:10.1006/jmbi.2000.4152.
- ↑ Murzin, Alexey G; Brenner, Steven E; Hubbard, Tim; Chothia, Cyrus (1995). "SCOP: a structural classification of proteins database for the investigation of sequences and structures". Journal of Molecular Biology 247 (4): 536-540. doi:10.1016/s0022-2836(05)80134-2.
- ↑ Orengo, Christine A; Michie, AD; Jones, S; Jones, David T; Swindells, MB; Thornton, Janet M (1997). "CATH-a hierarchic classification of protein domain structures". Structure 5 (8): 1093-1109. doi:10.1016/S0969-2126(97)00260-8.
- ↑ Wymer, Nathan; Buchanan, Louise V; Henderson, Darla; Mehta, Nupur; Botting, Catherine H; Pocivavsek, Luka; Fierke, Carol A; Toone, Eric J et al. (2001). "Directed evolution of a new catalytic site in 2-keto-3-deoxy-6-phosphogluconate aldolase from Escherichia coli". Structure 9 (1): 1-9. doi:10.1016/S0969-2126(00)00555-4.
- ↑ 45.0 45.1 Borman, Stu (2015). "Protein designers roll out a barrel". Chemical & Engineering News. Vol. 93, no. 47. p. 6. doi:10.1021/cen-09347-notw7.
- ↑ Goraj, Karine; Renard, Andre; Martial, Joseph A (1990). "Synthesis, purification and initial structural characterization of octarellin, a de novo polypeptide modelled on the α/β-barrel proteins". Protein Engineering, Design and Selection 3 (4): 259-266. doi:10.1093/protein/3.4.259.
- ↑ Beauregard, Marc; Goraj, Karine; Goffin, Vincent; Heremans, Karel; Goormaghtigh, Eric; Ruysschaert, Jean-Marie; Martial, Joseph A (1991). "Spectroscopic investigation of structure in octarellin (a de novo protein designed to adopt the α/β-barred packing)". Protein Engineering, Design and Selection 4 (7): 745-749. doi:10.1093/protein/4.7.745.
- ↑ Houbrechts, Annick; Moreau, Benoit; Abagyan, Ruben; Mainfroid, Veronique; Preaux, Gisele; Lamproye, Alain; Poncin, Alain; Goormaghtigh, Erik et al. (1995). "Second-generation octarellins: two new de novo (β/α)8 polypeptides designed for investigating the influence of β-residue packing on the α/β-barrel structure stability". Protein Engineering, Design and Selection 8 (3): 249-259. doi:10.1093/protein/8.3.249.
- ↑ Offredi, Fabrice; Dubail, Fabien; Kischel, Philippe; Sarinski, K; Stern, AS; Van de Weerdt, Cecile; Hoch, JC; Prosperi, Christelle et al. (2003). "De novo backbone and sequence design of an idealized α/β-barrel protein: evidence of stable tertiary structure". Journal of Molecular Biology 325 (1): 163-174. doi:10.1016/S0022-2836(02)01206-8.
- ↑ Figueroa, Maximiliano; Oliveira, Nicolas; Lejeune, Annabelle; Kaufmann, Kristian W; Dorr, Brent M; Matagne, Andre; Martial, Joseph A; Meiler, Jens et al. (2013). "Octarellin VI: Using rosetta to design a putative artificial (β/α)8 protein". PLoS ONE 8 (8): e71858. doi:10.1371/journal.pone.0071858.
- ↑ Figueroa, Maximiliano; Sleutel, Mike; Vandevenne, Marylene; Parvizi, Gregory; Attout, Sophie; Jacquin, Olivier; Vandenameele, Julie; Fischer, Axel W et al. (2016). "The unexpected structure of the designed protein Octarellin V.1 forms a challenge for protein structure prediction tools". Journal of Structural Biology 195 (1): 19-30. doi:10.1016/j.jsb.2016.05.004.
- ↑ Nanda, Vikas (2016). "Getting to the bottom of the TIM barrel". Nature Chemical Biology 12 (1): 2-3. doi:10.1038/nchembio.1987.
- ↑ Kaufmann, Kristian W; Lemmon, Gordon H; DeLuca, Samuel L; Sheehan, Jonathan H; Meiler, Jens (2010). "Practically useful: what the Rosetta protein modeling suite can do for you". Biochemistry 49 (14): 2987-2998. doi:10.1021/bi902153g.
- ↑ Koga, Nobuyasu; Tatsumi-Koga, Rie; Liu, Gaohua; Xiao, Rong; Acton, Thomas B; Montelione, Gaetano T; Baker, David (2012). "Principles for designing ideal protein structures". Nature 491 (7423): 222. doi:10.1038/nature11600.