WikiJournal Preprints/Leprosy
This article has been declined for publication by the WikiJournal of Medicine.
It was adapted from the Wikipedia page Leprosy and contains some or all of that page's content licensed under a CC BY-SA license.
It is archived below as a record. Discussion can be viewed here.
First submitted:
Declined:
Reviewer comments
QID: Q100400591
Suggested (provisional) preprint citation format:
Jennifer Dawson; Mary Olivia Hutton; Mayra May; James Heilman; Robert Dellavalle; et al.. "Leprosy". WikiJournal Preprints. Wikidata Q100400591.
License: ![]()
![]() This is an open access article distributed under the Creative Commons Attribution ShareAlike License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
This is an open access article distributed under the Creative Commons Attribution ShareAlike License, which permits unrestricted use, distribution, and reproduction, provided the original author and source are credited.
Roger Watson ![]() contact
contact
Gwinyai Masukume ![]()
Francisco Mayron Morais Soares ![]()
Article information
Abstract
Leprosy is spread between people, although extensive contact is necessary.[3][4] Leprosy has a low pathogenicity, and an estimated 95% of people who contract M. leprae do not develop the disease.[5] Spread is thought to occur through a cough or contact with fluid from the nose of a person infected by leprosy.[4][5] Genetic factors and immune function play a role in how easily a person catches the disease.[5][6] Leprosy does not spread during pregnancy to the unborn child, or through sexual contact.[4] Leprosy occurs more commonly among people living in poverty.[3] There are two main types of the disease – paucibacillary and multibacillary, which differ in the number of bacteria present.[3] A person with paucibacillary disease has five or fewer poorly pigmented numb skin patches while a person with multibacillary disease has more than five skin patches.[3] The diagnosis is confirmed by finding acid-fast bacilli in a biopsy of the skin.[3]
Leprosy is curable with multidrug therapy.[1] Treatment of paucibacillary leprosy is with the medications dapsone, rifampicin, and clofazimine for six months.[5] Treatment for multibacillary leprosy uses the same medications for 12 months.[5] A number of other antibiotics may also be used.[3] These treatments are provided free of charge by the World Health Organization.[1] People with leprosy can live with their families and go to school and work.[7] In 2018, there were 209,000 leprosy cases globally, down from 5.2 million in the 1980s.[8][9][10] The number of new cases in 2016 was 216,000.[1] Most new cases occur in 14 countries, with India accounting for more than half.[3][1] In the 20 years from 1994 to 2014, 16 million people worldwide were cured of leprosy.[1] About 200 cases per year are reported in the United States.[11]
Leprosy has affected humanity for thousands of years.[3] The disease takes its name from the Greek word λέπρᾱ (léprā), from λεπῐ́ς (lepís; "scale"), while the term "Hansen's disease" is named after the Norwegian physician Gerhard Armauer Hansen.[3] Leprosy has historically been associated with social stigma, which continues to be a barrier to self-reporting and early treatment.[1] Separating people affected by leprosy by placing them in leper colonies still occurs in some areas of India,[12] China,[13] Africa,[14] and Thailand.[15] Most colonies have closed, as leprosy is not very contagious.[14] Some consider the word "leper" offensive, preferring the phrase "person affected with leprosy".[16] Leprosy is classified as a neglected tropical disease.[17] World Leprosy Day was started in 1954 to draw awareness to those affected by leprosy.[18]
Signs and symptoms
[edit | edit source]Common symptoms present in the different types of leprosy include a runny nose; dry scalp; eye problems; skin lesions; muscle weakness; reddish skin; smooth, shiny, diffuse thickening of facial skin, ear, and hand; loss of sensation in fingers and toes; thickening of peripheral nerves; a flat nose due to destruction of nasal cartilage; and changes in phonation and other aspects of speech production.[19][20] In addition, atrophy of the testes and impotence may occur.[21]
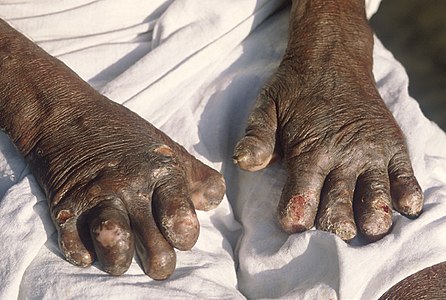
Leprosy can affect people in different ways.[5] The average incubation period is 5 years.[1] People may begin to notice symptoms within the first year or up to 20 years after infection.[1] The first noticeable sign of leprosy is often the development of pale or pink coloured patches of skin that may be insensitive to temperature or pain.[22] Patches of discolored skin are sometimes accompanied or preceded by nerve problems including numbness or tenderness in the hands or feet.[22][23] Secondary infections (additional bacterial or viral infections) can result in tissue loss, causing fingers and toes to become shortened and deformed, as cartilage is absorbed into the body.[24][25] A person's immune response differs depending on the form of leprosy.[26]
Approximately 30% of people affected with leprosy experience nerve damage.[27] The nerve damage sustained is reversible when treated early, but becomes permanent when appropriate treatment is delayed by several months. Damage to nerves may cause loss of muscle function, leading to paralysis. It may also lead to sensation abnormalities or numbness, which may lead to additional infections, ulcerations, and joint deformities.[27]
-
Paucibacillary leprosy (PB): Pale skin patch with loss of sensation
-
Skin lesions on the thigh of a person with leprosy
-
Hands deformed by leprosy
Cause
[edit | edit source]M. leprae and M. lepromatosis
[edit | edit source]
M. leprae and M. lepromatosis are the mycobacteria that cause leprosy.[27] M. lepromatosis is a relatively newly identified mycobacterium isolated from a fatal case of diffuse lepromatous leprosy in 2008.[28][29] M. lepromatosis is indistinguishable clinically from M. leprae.[30]
M. leprae is an intracellular, acid-fast bacterium that is aerobic and rod-shaped.[31] M. leprae is surrounded by the waxy cell envelope coating characteristic of the genus Mycobacterium.[31]
Genetically, M. leprae and M. lepromatosis lack the genes that are necessary for independent growth.[32] M. leprae and M. lepromatosis are obligate intracellular pathogens, and can not be grown (cultured) in the laboratory.[32] The inability to culture M. leprae and M. lepromatosis has resulted in a difficulty definitively identifying the bacterial organism under a strict interpretation of Koch's postulates.[28][32]
While the causative organisms have to date been impossible to culture in vitro, it has been possible to grow them in animals such as mice and armadillos.[33][34]
Naturally occurring infection has been reported in nonhuman primates (including the African chimpanzee, the sooty mangabey, and the cynomolgus macaque), armadillos,[35] and red squirrels.[36] Multilocus sequence typing of the armadillo M. leprae strains suggests that they were of human origin for at most a few hundred years.[37] Thus, it is suspected that armadillos first acquired the organism incidentally from early American explorers.[38] This incidental transmission was sustained in the armadillo population, and it may be transmitted back to humans, making leprosy a zoonotic disease (spread between humans and animals).[38]
Red squirrels (Sciurus vulgaris), a threatened species in Great Britain, were found to carry leprosy in November 2016.[39] It has been suggested that the trade in red squirrel fur, highly prized in the medieval period and intensively traded, may have been responsible for the leprosy epidemic in medieval Europe.[40] A pre-Norman-era skull excavated in Hoxne, Suffolk, in 2017 was found to carry DNA from a strain of Mycobacterium leprae, which closely matched the strain carried by modern red squirrels on Brownsea Island, UK.[40][41]
Risk factors
[edit | edit source]The greatest risk factor for developing leprosy is contact with another person infected by leprosy.[1] People who are exposed to a person who has leprosy are 5–8 times more likely to develop leprosy than members of the general population.[42] Leprosy also occurs more commonly among those living in poverty.[3] Not all people who are infected with M. leprae develop symptoms.[43][44]
Conditions that reduce immune function, such as malnutrition, other illnesses, or genetic mutations, may increase the risk of developing leprosy.[42] Infection with HIV does not appear to increase the risk of developing leprosy.[45] Certain genetic factors in the person exposed have been associated with developing lepromatous or tuberculoid leprosy.[46]
Transmission
[edit | edit source]Transmission of leprosy occurs during close contact with those who are infected.[1] Transmission of leprosy is through the upper respiratory tract.[5][47] Older research suggested the skin as the main route of transmission, but recent research has increasingly favored the respiratory route.[48]
Leprosy is not sexually transmitted and is not spread through pregnancy to the unborn child.[1][4] The majority (95%) of people who are exposed to M. Leprae do not develop leprosy; casual contact such as shaking hands and sitting next to someone with leprosy does not lead to transmission.[1][49] People are considered non-infectious 72 hours after starting appropriate multi-drug therapy.[50]
Two exit routes of M. leprae from the human body often described are the skin and the nasal mucosa, although their relative importance is not clear. Lepromatous cases show large numbers of organisms deep in the dermis, but whether they reach the skin surface in sufficient numbers is doubtful.[51]
Leprosy may also be transmitted to humans by armadillos, although the mechanism is not fully understood.[4][52][53]
Genetics
[edit | edit source]| Name | Locus | OMIM | Gene |
|---|---|---|---|
| LPRS1 | 10p13 | Online 'Mendelian Inheritance in Man' (OMIM) 609888 | |
| LPRS2 | 6q25 | Online 'Mendelian Inheritance in Man' (OMIM) 607572 | PARK2, PACRG |
| LPRS3 | 4q32 | Online 'Mendelian Inheritance in Man' (OMIM) 246300 | TLR2 |
| LPRS4 | 6p21.3 | Online 'Mendelian Inheritance in Man' (OMIM) 610988 | LTA |
| LPRS5 | 4p14 | Online 'Mendelian Inheritance in Man' (OMIM) 613223 | TLR1 |
| LPRS6 | 13q14.11 | Online 'Mendelian Inheritance in Man' (OMIM) 613407 |
Not all people who are infected or exposed to M. leprae develop leprosy, and genetic factors are suspected to play a role in susceptibility to an infection.[54] Cases of leprosy often cluster in families and several genetic variants have been identified.[54] In many people who are exposed, the immune system is able to eliminate the leprosy bacteria during the early infection stage before severe symptoms develop.[55] A genetic defect in cell-mediated immunity may cause a person to be susceptible to develop leprosy symptoms after exposure to the bacteria.[56] The region of DNA responsible for this variability is also involved in Parkinson's disease, giving rise to current speculation that the two disorders may be linked at the biochemical level.[56]
Mechanism
[edit | edit source]Most leprosy complications are the result of nerve damage. The nerve damage occurs due to direct invasion by the M. leprae bacteria and a person's immune response resulting in inflammation.[27] The molecular mechanism underlying how M. leprae produces the symptoms of leprosy is not clear,[10] but M. leprae has been shown to bind to Schwann cells, which may lead to nerve injury including demyelination and a loss of nerve function (specifically a loss of axonal conductance).[57] Numerous molecular mechanisms have been associated with this nerve damage including the presence of a laminin-binding protein and the glycoconjugate (PGL-1) on the surface of M. leprae that can bind to laminin on peripheral nerves.[57]
As part of the human immune response, white blood cell-derived macrophages may engulf M. leprae by phagocytosis.[57]
In the initial stages, small sensory and autonomic nerve fibers in the skin of a person with leprosy are damaged.[27] This damage usually results in hair loss to the area, a loss of the ability to sweat, and numbness (decreased ability to detect sensations such as temperature and touch). Further peripheral nerve damage may result in skin dryness, more numbness, and muscle weaknesses or paralysis in the area affected.[27] The skin can crack and if the skin injuries are not carefully cared for, there is a risk for a secondary infection that can lead to more severe damage.[27]
Diagnosis
[edit | edit source]
In countries where people are frequently infected, a person is considered to have leprosy if they one of the following two signs:
Skin lesions can be single or many, and usually hypopigmented, although occasionally reddish or copper-colored.[58] The lesions may be flat (macules), raised (papules), or solid elevated areas (nodular).[58] Experiencing sensory loss at the skin lesion is a feature that can help determine if the lesion is caused by leprosy or if the lesion is caused by another disorder such as tinea versicolor.[58][59] Thickened nerves are associated with leprosy and can be accompanied by loss of sensation or muscle weakness, but without the characteristic skin lesion and sensory loss, muscle weakness is not considered a reliable sign of leprosy.[58]
In some cases, acid-fast leprosy bacilli in skin smears are considered diagnostic; however, the diagnosis is typically made without laboratory tests, based on symptoms.[58] If a person has a new leprosy diagnosis and already has a visible disability due to leprosy, the diagnosis is considered late.[27]
In countries or areas where leprosy is uncommon, such as the United States, diagnosis of leprosy is often delayed because healthcare providers are unaware of leprosy and its symptoms.[60] Early diagnosis and treatment prevent nerve involvement, the hallmark of leprosy, and the disability it causes.[1][60]
There is no recommended test to diagnose latent leprosy in people without symptoms.[5] Few people with latent leprosy test positive for anti PGL-1.[43] The presence of M. leprae bacterial DNA can be identified using a polymerase chain reaction (PCR)-based technique.[61] This molecular test alone is not sufficient to diagnose a person, but this approach may be used to identify someone who is at high risk of developing or transmitting leprosy such as those with few lesions or an atypical clinical presentation.[61][62]
Classification
[edit | edit source]Several different approaches for classifying leprosy exist. There are similarities between the classification approaches.
- The World Health Organization system distinguishes "paucibacillary" and "multibacillary" based upon the proliferation of bacteria.[63] ("pauci-" refers to a low quantity.)
- The Ridley-Jopling scale provides five gradations.[64][65][66]
- The ICD-10, though developed by the WHO, uses Ridley-Jopling and not the WHO system. It also adds an indeterminate ("I") entry.[51]
- In MeSH, three groupings are used.
| WHO | Ridley-Jopling | ICD-10 | MeSH | Description | Lepromin test |
|---|---|---|---|---|---|
| Paucibacillary | tuberculoid ("TT"), borderline tuberculoid ("BT") |
A30.1, A30.2 | Tuberculoid | It is characterized by one or more hypopigmented skin macules and patches where skin sensations are lost because of damaged peripheral nerves that have been attacked by the human host's immune cells. TT is characterized by the formation of epithelioid cell granulomas with a large number of epithelioid cells. In this form of leprosy Mycobacterium leprae are either absent from the lesion or occur in very small numbers. This type of leprosy is most benign.[67][68] | Positive |
| Multibacillary | midborderline or borderline ("BB") |
A30.3 | Borderline | Borderline leprosy is of intermediate severity and is the most common form. Skin lesions resemble tuberculoid leprosy, but are more numerous and irregular; large patches may affect a whole limb, and peripheral nerve involvement with weakness and loss of sensation is common. This type is unstable and may become more like lepromatous leprosy or may undergo a reversal reaction, becoming more like the tuberculoid form. | |
| Multibacillary | borderline lepromatous ("BL"), and lepromatous ("LL") |
A30.4, A30.5 | Lepromatous | It is associated with symmetric skin lesions, nodules, plaques, thickened dermis, and frequent involvement of the nasal mucosa resulting in nasal congestion and nose bleeds, but, typically, detectable nerve damage is late. Loss of eyebrows and lashes can be seen in advanced disease.[69] LL is characterized by the absence of epithelioid cells in the lesions. In this form of leprosy Mycobacterium leprae are found in lesion in large numbers. This is the most unfavorable clinical variant of leprosy, which occurs with a generalized lesion of the skin, mucous membranes, eyes, peripheral nerves, lymph nodes, and internal organs.[67][68] | Negative |
Leprosy may also occur with only neural involvement, without skin lesions.[1][70][71][72][73][74]
Prevention
[edit | edit source]Early detection of the disease is important, since physical and neurological damage may be irreversible even if cured.[1] Medications can decrease the risk of those living with people who have leprosy from acquiring the disease and likely those with whom people with leprosy come into contact outside the home.[10] The WHO recommends that preventive medicine be given to people who are in close contact with someone who has leprosy.[5] The suggested preventive treatment is a single dose of rifampicin (SDR) in adults and children over 2 years old who do not already have leprosy or tuberculosis.[5] Preventive treatment is associated with a 57% reduction in infections within 2 years and a 30% reduction in infections within 6 years.[5]
The Bacillus Calmette–Guérin (BCG) vaccine offers a variable amount of protection against leprosy in addition to its target of tuberculosis.[75] It appears to be 26% to 41% effective (based on controlled trials) and about 60% effective based on observational studies with two doses possibly working better than one.[76][77] The WHO concluded in 2018 that the BCG vaccine at birth reduces leprosy risk and is recommended for people who live in countries with high incidence of TB and leprosy.[78] People living in the same home as a person with leprosy are suggested to take a BCG booster which may improve their immunity by 56%.[79][80] Development of a more effective vaccine is ongoing.[10][81][82][83]
Treatment
[edit | edit source]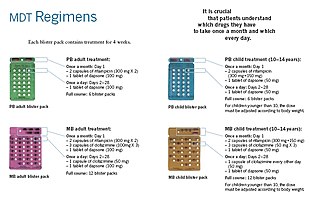
Anti-leprosy medication
[edit | edit source]A number of leprostatic agents are available for treatment. A 3-drug regimen of rifampicin, dapsone and clofazimine is recommended for all people with leprosy, for 6 months for paucibacillary leprosy and 12 months for multibacillary leprosy.[5]
Multidrug therapy (MDT) remains highly effective, and people are no longer infectious after the first monthly dose.[1] It is safe and easy to use under field conditions due to its presentation in calendar blister packs.[1] Post-treatment relapse rates remain low.[1] Resistance has been reported in several countries, although the number of cases is small.[84] People with rifampicin-resistant leprosy may be treated with second line drugs such as fluoroquinolones, minocycline, or clarithromycin, but the treatment duration is 24 months due to their lower bactericidal activity.[85] Evidence on the potential benefits and harms of alternative regimens for drug-resistant leprosy is not yet available.[5]
Skin changes
[edit | edit source]For people with nerve damage, protective footwear may help prevent ulcers and secondary infection.[27] Canvas shoes may be better than PVC-boots.[27] There may be no difference between double rocker shoes and below-knee plaster.[27]
Topical ketanserin seems to have a better effect on ulcer healing than clioquinol cream or zinc paste, but the evidence for this is weak.[27] Phenytoin applied to the skin improves skin changes to a greater degree when compared to saline dressings.[27]
Outcomes
[edit | edit source]Leprosy is curable, however, when left untreated leprosy can cause permanent physical impairments and damage to a person's nerves, skin, eyes, and limbs.[86] Despite leprosy not being very infectious and having a low pathogenicity, there is still significant stigma and prejudice associated with the disease.[87] Due to this stigma, leprosy can effect a person's participation in social activities and may also effect the lives of their family and friends.[87] People with leprosy are also at a higher risk for problems with their mental well being.[87] The social stigma may contribute to problems obtaining employment, financial difficulties, and social isolation.[87] Efforts to reduce discrimination and improve the stigma surrounding leprosy may help improve outcomes for people with leprosy.[88]
Epidemiology
[edit | edit source]

In 2018, there were 208,619 new cases of leprosy recorded, a slight decrease from 2017.[92] In 2015, 94% of the new leprosy cases were confined to 14 countries.[93] India reported the greatest number of new cases (60% of reported cases), followed by Brazil (13%) and Indonesia (8%).[93] Although the number of cases worldwide continues to fall, there are parts of the world where leprosy is more common, including Brazil, South Asia (India, Nepal, Bhutan), some parts of Africa (Tanzania, Madagascar, Mozambique), and the western Pacific.[93] About 150 to 250 cases are diagnosed in the United States each year.[94]
In the 1960s, there were tens of millions of leprosy cases recorded when the bacteria started to develop resistance to dapsone, the most common treatment option at the time.[1][10] International (e.g., the WHO's "Global Strategy for Reducing Disease Burden Due to Leprosy") and national (e.g., the International Federation of Anti-Leprosy Associations) initiatives have reduced the total number and the number of new cases of the disease.[10][95]
Disease burden
[edit | edit source]The number of new leprosy cases is difficult to measure and monitor due to leprosy's long incubation period, delays in diagnosis after onset of the disease, and lack of medical care in affected areas.[96] The registered prevalence of the disease is used to determine disease burden.[97] Registered prevalence is a useful proxy indicator of the disease burden, as it reflects the number of active leprosy cases diagnosed with the disease and receiving treatment with MDT at a given point in time.[97] The prevalence rate is defined as the number of cases registered for MDT treatment among the population in which the cases have occurred, again at a given point in time.[97]
Public policy
[edit | edit source]A goal of the World Health Organization is to "eliminate leprosy" and in 2016 the organization launched "Global Leprosy Strategy 2016–2020: Accelerating towards a leprosy-free world".[98] Elimination of leprosy is defined as "reducing the proportion of leprosy patients in the community to very low levels, specifically to below one case per 10 000 population".[99] Diagnosis and treatment with multidrug therapy are effective, and a 45% decline in disease burden has occurred since multidrug therapy has become more widely available.[100] The organization emphasizes the importance of fully integrating leprosy treatment into public health services, effective diagnosis and treatment, and access to information.[100] The approach includes supporting an increase in health care professionals who understand the disease, and a coordinated and renewed political commitment that includes coordination between countries and improvements in the methodology for collecting and analysing data.[98]
Interventions in the "Global Leprosy Strategy 2016–2020: Accelerating towards a leprosy-free world":[98]
[edit | edit source]- Early detection of cases focusing on children with the aim to reduce transmission and disabilities
- Enhanced healthcare services and improved access for people who may be marginalized
- For countries where leprosy is an endemic, further interventions include: improved screening of close contacts, improved treatment regimen's, and interventions to reduce stigma and discrimination against people who have leprosy.
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 "Leprosy Fact sheet N°101". World Health Organization. January 2014. Archived from the original on 2013-12-12.
- ↑ Sotiriou, MC; Stryjewska, BM; Hill, C (7 September 2016). "Two Cases of Leprosy in Siblings Caused by Mycobacterium lepromatosis and Review of the Literature.". The American Journal of Tropical Medicine and Hygiene 95 (3): 522–7. doi:10.4269/ajtmh.16-0076. PMID 27402522. PMC 5014252. //www.ncbi.nlm.nih.gov/pmc/articles/PMC5014252/.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 "Current status of leprosy: epidemiology, basic science and clinical perspectives.". The Journal of Dermatology 39 (2): 121–9. February 2012. doi:10.1111/j.1346-8138.2011.01370.x. PMID 21973237.
- ↑ 4.0 4.1 4.2 4.3 4.4 "Hansen's Disease (Leprosy) Transmission". cdc.gov. April 29, 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 Guidelines for the Diagnosis, Treatment and Prevention of Leprosy. World Health Organization. 2018. pp. xiii. ISBN 978-92-9022-638-3. https://apps.who.int/iris/bitstream/handle/10665/274127/9789290226383-eng.pdf?ua=1.
- ↑ Montoya, D; Modlin, RL (2010). "Learning from leprosy: insight into the human innate immune response.". Advances in Immunology 105: 1–24. doi:10.1016/S0065-2776(10)05001-7. ISBN 9780123813022. PMID 20510728.
- ↑ CDC (2018-01-26). "World Leprosy Day". Centers for Disease Control and Prevention. Retrieved 2019-07-04.
- ↑ "Leprosy". www.who.int. Retrieved 10 February 2020.
- ↑ "Global leprosy situation, 2012". Wkly. Epidemiol. Rec. 87 (34): 317–28. August 2012. PMID 22919737.
- ↑ 10.0 10.1 10.2 10.3 10.4 10.5 Rodrigues LC; Lockwood DNj (June 2011). "Leprosy now: epidemiology, progress, challenges, and research gaps.". The Lancet Infectious Diseases 11 (6): 464–70. doi:10.1016/S1473-3099(11)70006-8. PMID 21616456.
- ↑ "Hansen's Disease Data & Statistics". Health Resources and Services Administration. Archived from the original on 4 January 2015. Retrieved 12 January 2015.
- ↑ Walsh F (2007-03-31). "The hidden suffering of India's lepers". BBC News. Archived from the original on 2007-05-29.
- ↑ Lyn TE (2006-09-13). "Ignorance breeds leper colonies in China". Independat News & Media. Archived from the original on 2010-04-08. Retrieved 2010-01-31.
- ↑ 14.0 14.1 Byrne, Joseph P. (2008). Encyclopedia of pestilence, pandemics, and plagues. Westport, Conn.[u.a.]: Greenwood Press. p. 351. ISBN 978-0-313-34102-1. https://archive.org/details/encyclopediaofpe00jose_0.
- ↑ Pisuthipan, Arusa (6 July 2020). "Forgotten victims of the virus". Bangkok Post. Retrieved 6 July 2020.
- ↑ Leprosy a practical guide. Milan: Springer. 2012. p. 326. ISBN 978-88-470-2376-5. https://books.google.com/books?id=ZQ2k0xgIph4C&pg=PA326.
- ↑ "Neglected Tropical Diseases". cdc.gov. June 6, 2011. Archived from the original on 4 December 2014. Retrieved 28 November 2014.
- ↑ McMenamin, Dorothy (2011). Leprosy and stigma in the South Pacific: a region-by-region history with first person accounts. Jefferson, N.C.: McFarland. p. 17. ISBN 978-0-7864-6323-7. https://books.google.com/books?id=lZPvQTJ8SE0C&pg=PA17.
- ↑ "Signs and Symptoms | Hansen's Disease (Leprosy) | CDC". www.cdc.gov. 2018-10-22. Retrieved 2019-07-22.
- ↑ "Leprosy Specific Orofacial Aspects". ResearchGate. Retrieved 2019-07-22.
- ↑ admin (2016-02-11). "Pathogenesis and Pathology of Leprosy". International Textbook of Leprosy. Retrieved 2019-07-22.
- ↑ 22.0 22.1 WHO Expert Committee on Leprosy - Eight report. World Health Organization. 2012. pp. 11–12. ISBN 9789241209687. http://www.searo.who.int/entity/global_leprosy_programme/publications/8th_expert_comm_2012.pdf. Retrieved 9 May 2018.
- ↑ "Clinical aspects of leprosy". Clinics in Dermatology 33 (1): 26–37. 2015. doi:10.1016/j.clindermatol.2014.07.002. PMID 25432808.
- ↑ Kulkarni GS (2008). Textbook of Orthopedics and Trauma (2nd ed.). Jaypee Brothers Publishers. p. 779. ISBN 978-81-8448-242-3.
- ↑ "Q and A about leprosy". American Leprosy Missions. Archived from the original on 2012-10-04. Retrieved 2011-01-22.
Do fingers and toes fall off when someone gets leprosy? No. The bacillus attacks nerve endings and destroys the body's ability to feel pain and injury. Without feeling pain, people injure themselves on fire, thorns, rocks, even hot coffee cups. Injuries become infected and result in tissue loss. Fingers and toes become shortened and deformed as the cartilage is absorbed into the body.
- ↑ de Sousa, Jorge Rodrigues; Sotto, Mirian Nacagami; Simões Quaresma, Juarez Antonio (2017). "Leprosy As a Complex Infection: Breakdown of the Th1 and Th2 Immune Paradigm in the Immunopathogenesis of the Disease". Frontiers in Immunology 8: 1635. doi:10.3389/fimmu.2017.01635. ISSN 1664-3224. PMID 29234318. PMC 5712391. //www.ncbi.nlm.nih.gov/pmc/articles/PMC5712391/.
- ↑ 27.00 27.01 27.02 27.03 27.04 27.05 27.06 27.07 27.08 27.09 27.10 27.11 27.12 Reinar, Liv Merete; Forsetlund, Louise; Lehman, Linda Faye; Brurberg, Kjetil G (2019-07-31). Cochrane Skin Group. ed. "Interventions for ulceration and other skin changes caused by nerve damage in leprosy". Cochrane Database of Systematic Reviews 7: CD012235. doi:10.1002/14651858.CD012235.pub2. PMID 31425632. PMC 6699662. //www.ncbi.nlm.nih.gov/pmc/articles/PMC6699662/.
- ↑ 28.0 28.1 "New Leprosy Bacterium: Scientists Use Genetic Fingerprint To Nail 'Killing Organism'". ScienceDaily. 2008-11-28. Archived from the original on 2010-03-13. Retrieved 2010-01-31.
- ↑ Ryan, Kenneth J.; Ray, C. George, eds (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. pp. 451–3. ISBN 978-0-8385-8529-0. OCLC 61405904. https://archive.org/details/sherrismedicalmi00ryan.
- ↑ "Genomics Insights into the Biology and Evolution of Leprosy Bacilli". International Textbook of Leprosy. 2016-02-11. Retrieved 2019-02-11.
- ↑ 31.0 31.1 McMurray DN (1996). "Mycobacteria and Nocardia". Baron's Medical Microbiology (4th ed.). Univ of Texas Medical Branch. ISBN 978-0-9631172-1-2. OCLC 33838234. https://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=mmed.section.1833.
- ↑ 32.0 32.1 32.2 "Uncultivable bacteria: Implications and recent trends towards identification". Indian Journal of Medical Microbiology 20 (4): 174–7. 1 October 2002. PMID 17657065. Archived from the original on 27 September 2007. http://www.ijmm.org/article.asp?issn=0255-0857;year=2002;volume=20;issue=4;spage=174;epage=177;aulast=Bhattacharya. Retrieved 20 August 2007.
- ↑ "WHO | Microbiology: culture in vitro". WHO. Retrieved 2019-07-22.
- ↑ admin (2016-02-11). "The Armadillo Model for Leprosy". International Textbook of Leprosy. Retrieved 2019-07-22.
- ↑ Loughry WJ, Truman RW, McDonough CM, Tilak MK, Garnier S, et al. (2009) "Is leprosy spreading among nine-banded armadillos in the southeastern United States?" J Wildl Dis 45: 144–152.
- ↑ Meredith, Anna; Del Pozo, Jorge; Smith, Sionagh; Milne, Elspeth; Stevenson, Karen; McLuckie, Joyce (September 2014). "Leprosy in red squirrels in Scotland". Veterinary Record 175 (11): 285–286. doi:10.1136/vr.g5680. PMID 25234460.
- ↑ Monot M, Honoré N, Garnier T, Araoz R, Coppee JY, et al. (2005). "On the origin of leprosy". Science 308: 1040–1042.
- ↑ 38.0 38.1 Han, Xiang Y.; Silva, Francisco J.; Baker, Stephen (13 February 2014). "On the Age of Leprosy". PLOS Neglected Tropical Diseases 8 (2): e2544. doi:10.1371/journal.pntd.0002544. PMID 24551248. PMC 3923669. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3923669/.
- ↑ "Red squirrels in the British Isles are infected with leprosy bacilli", Dr. Andrej Benjak, Prof Anna Meredith and others. Science, 11 November 2016. Archived 11 November 2016 at the Wayback Machine. Retrieved 11 November 2016.
- ↑ 40.0 40.1 "Could squirrel fur trade have contributed to England's medieval leprosy outbreak?". ScienceDaily. Retrieved 21 November 2018.
- ↑ Inskip, S; Taylor, GM; Anderson, S; Stewart, G (November 2017). "Leprosy in pre-Norman Suffolk, UK: biomolecular and geochemical analysis of the woman from Hoxne". Journal of Medical Microbiology 66 (11): 1640–1649. doi:10.1099/jmm.0.000606. PMID 28984227. http://epubs.surrey.ac.uk/846050/1/Inskip%20et%20al%20submission%20proof%20%28002%29.pdf.
- ↑ 42.0 42.1 Schreuder, P.A.M.; Noto, S.; Richardus J.H. (January 2016). "Epidemiologic trends of leprosy for the 21st century". Clinics in Dermatology 34 (1): 24–31. doi:10.1016/j.clindermatol.2015.11.001. PMID 26773620.
- ↑ 43.0 43.1 Penna, Maria Lucia F.; Penna, Gerson O.; Iglesias, Paula C.; Natal, Sonia; Rodrigues, Laura C. (2016-05-18). Small, Pamela L. C.. ed. "Anti-PGL-1 Positivity as a Risk Marker for the Development of Leprosy among Contacts of Leprosy Cases: Systematic Review and Meta-analysis". PLOS Neglected Tropical Diseases 10 (5): e0004703. doi:10.1371/journal.pntd.0004703. ISSN 1935-2735. PMID 27192199. PMC 4871561. //www.ncbi.nlm.nih.gov/pmc/articles/PMC4871561/.
- ↑ "Genetic dissection of immunity in leprosy". Curr. Opin. Immunol. 17 (1): 44–8. 2005. doi:10.1016/j.coi.2004.11.006. PMID 15653309.
- ↑ "Human immunodeficiency virus and leprosy: an update.". Dermatologic Clinics 29 (1): 125–8. January 2011. doi:10.1016/j.det.2010.08.016. PMID 21095536.
- ↑ admin (2016-02-11). "Epidemiology of Leprosy". International Textbook of Leprosy. Retrieved 2019-07-30.
- ↑ Chavarro-Portillo, Bibiana; Soto, Carlos Yesid; Guerrero, Martha Inírida (September 2019). "Mycobacterium leprae's evolution and environmental adaptation". Acta Tropica 197: 105041. doi:10.1016/j.actatropica.2019.105041. ISSN 1873-6254. PMID 31152726.
- ↑ Eichelmann, K.; González González, S. E.; Salas-Alanis, J. C.; Ocampo-Candiani, J. (2013-09-01). "Leprosy. An Update: Definition, Pathogenesis, Classification, Diagnosis, and Treatment". Actas Dermo-Sifiliográficas (English Edition) 104 (7): 554–563. doi:10.1016/j.adengl.2012.03.028. ISSN 1578-2190. PMID 23870850.
- ↑ "Hansen's Disease (Leprosy) Transmission". cdc.gov. April 29, 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- ↑ Lockwood, Diana N J; Kumar, Bhushan (2004-06-19). "Treatment of leprosy". BMJ : British Medical Journal 328 (7454): 1447–1448. doi:10.1136/bmj.328.7454.1447. ISSN 0959-8138. PMID 15205269. PMC 428501. //www.ncbi.nlm.nih.gov/pmc/articles/PMC428501/.
- ↑ 51.0 51.1 "What Is Leprosy?" THE MEDICAL NEWS | from News-Medical.Net – Latest Medical News and Research from Around the World. Web. 20 Nov. 2010. "What is Leprosy?". 2009-11-18. Archived from the original on 2013-06-06. Retrieved 2013-05-14..
- ↑ "Probable Zoonotic Leprosy in the Southern United States". The New England Journal of Medicine (Massachusetts Medical Society) 364 (17): 1626–1633. April 2011. doi:10.1056/NEJMoa1010536. PMID 21524213. PMC 3138484. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3138484/.
- ↑ "Hansen's Disease (Leprosy) Transmission". cdc.gov. April 29, 2013. Archived from the original on 13 March 2015. Retrieved 28 February 2015.
- ↑ 54.0 54.1 Cambri, Geison; Mira, Marcelo Távora (2018-07-20). "Genetic Susceptibility to Leprosy—From Classic Immune-Related Candidate Genes to Hypothesis-Free, Whole Genome Approaches". Frontiers in Immunology 9: 1674. doi:10.3389/fimmu.2018.01674. ISSN 1664-3224. PMID 30079069. PMC 6062607. //www.ncbi.nlm.nih.gov/pmc/articles/PMC6062607/.
- ↑ Cook, Gordon C. (2009). Manson's tropical diseases. (22nd ed.). [Edinburgh]: Saunders. p. 1056. ISBN 978-1-4160-4470-3. https://books.google.com/books?id=CF2INI0O6l0C&pg=PA1056.
- ↑ 56.0 56.1 "Linkage of leprosy susceptibility to Parkinson's disease genes". International Journal of Leprosy and Other Mycobacterial Diseases 72 (2): 169–70. Jun 2004. doi:10.1489/1544-581X(2004)072<0169:LOLSTP>2.0.CO;2. ISSN 0148-916X. PMID 15301585. Archived from the original on January 5, 2012. http://www.leprosy-ila.org/leprosyjournal/pdf/72/i0148-916X-72-2.pdf. Retrieved January 31, 2011.
- ↑ 57.0 57.1 57.2 Bhat, Ramesh Marne; Prakash, Chaitra (2012). "Leprosy: An Overview of Pathophysiology". Interdisciplinary Perspectives on Infectious Diseases 2012: 181089. doi:10.1155/2012/181089. ISSN 1687-708X. PMID 22988457. PMC 3440852. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3440852/.
- ↑ 58.0 58.1 58.2 58.3 58.4 58.5 58.6 "Diagnosis of Leprosy." WHO. from "WHO | Diagnosis of leprosy". Archived from the original on 2014-06-05. Retrieved 2014-07-14. accessed on 14 July 2014.
- ↑ Moschella, Samuel L; Garcia-Albea, Victoria (September 2016). "International Textbook of Leprosy" (PDF). Differential Diagnosis of Leprosy. p. 3, Section 2.3. Retrieved July 4, 2019.
- ↑ 60.0 60.1 U.S. Department of Health and Human Services, Health Resources and Services Administration. (n.d.). National Hansen's disease (leprosy) program Retrieved from "National Hansen's Disease (Leprosy) Program". Archived from the original on 2011-02-10. Retrieved 2013-05-12.
- ↑ 61.0 61.1 Martinez, Alejandra Nóbrega; Talhari, Carolina; Moraes, Milton Ozório; Talhari, Sinésio (2014). "PCR-based techniques for leprosy diagnosis: from the laboratory to the clinic". PLOS Neglected Tropical Diseases 8 (4): e2655. doi:10.1371/journal.pntd.0002655. ISSN 1935-2735. PMID 24722358. PMC 3983108. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3983108/.
- ↑ Tatipally, Sushma; Srikantam, Aparna; Kasetty, Sanjay (2018-10-01). "Polymerase Chain Reaction (PCR) as a Potential Point of Care Laboratory Test for Leprosy Diagnosis—A Systematic Review". Tropical Medicine and Infectious Disease 3 (4): 107. doi:10.3390/tropicalmed3040107. ISSN 2414-6366. PMID 30275432. PMC 6306935. //www.ncbi.nlm.nih.gov/pmc/articles/PMC6306935/.
- ↑ Smith DS (2008-08-19). "Leprosy: Overview". eMedicine Infectious Diseases. Archived from the original on 2010-02-18. Retrieved 2010-02-01.
- ↑ "Pitfalls in the cytological classification of borderline leprosy in the Ridley-Jopling scale". Diagn. Cytopathol. 30 (6): 386–8. June 2004. doi:10.1002/dc.20012. PMID 15176024.
- ↑ Ridley DS, Jopling WH; Jopling (1966). "Classification of leprosy according to immunity. A five-group system". Int. J. Lepr. Other Mycobact. Dis. 34 (3): 255–73. PMID 5950347.
- ↑ James, William D.Expression error: Unrecognized word "etal". (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. pp. 344–346. ISBN 978-0-7216-2921-6. https://archive.org/details/andrewsdiseasess00mdwi_659.
- ↑ 67.0 67.1 "Leprosy: an overview of pathophysiology". Interdiscip Perspect Infect Dis 2012: 1–6. 2012. doi:10.1155/2012/181089. PMID 22988457.
- ↑ 68.0 68.1 "Leprosy: a review of laboratory and therapeutic aspects". An Bras Dermatol 89 (3): 389–401. 2014. doi:10.1590/abd1806-4841.20142460. PMID 24937811. PMC 4056695. //www.ncbi.nlm.nih.gov/pmc/articles/PMC4056695/.
- ↑ Kumar, Bhushan; Uprety, Shraddha; Dogra, Sunil (2016-02-11). "Clinical Diagnosis of Leprosy". International Textbook of Leprosy. Retrieved 2019-02-12.
- ↑ "Criteria for diagnosis of pure neural leprosy". J. Neurol. 250 (7): 806–9. July 2003. doi:10.1007/s00415-003-1081-5. PMID 12883921.
- ↑ "Primary neuritic leprosy: a reappraisal at a tertiary care hospital". Indian J Lepr 78 (3): 261–7. 2006. PMID 17120509.
- ↑ "Three cases of pure neuritic (PN) leprosy at detection in which skin lesions became visible during their course". Nihon Hansenbyo Gakkai Zasshi 69 (2): 101–6. July 2000. doi:10.5025/hansen.69.101. PMID 10979277.
- ↑ "Neuritic leprosy: further progression and significance". Acta Leprol 9 (4): 187–94. 1995. PMID 8711979.
- ↑ "Neuritic leprosy: epidemiology and therapeutic responsiveness". Lepr Rev 63 (3): 263–8. September 1992. doi:10.5935/0305-7518.19920031. PMID 1406021.
- ↑ "Advances and hurdles on the way toward a leprosy vaccine". Hum Vaccin 7 (11): 1172–83. November 2011. doi:10.4161/hv.7.11.16848. PMID 22048122. PMC 3323495. //www.ncbi.nlm.nih.gov/pmc/articles/PMC3323495/.
- ↑ "The role of BCG in prevention of leprosy: a meta-analysis". Lancet Infect Dis 6 (3): 162–70. March 2006. doi:10.1016/S1473-3099(06)70412-1. PMID 16500597.
- ↑ "BCG vaccination and leprosy protection: Review of current evidence and status of BCG in leprosy control". Expert Review of Vaccines 9 (2): 209–222. 2010. doi:10.1586/ERV.09.161. PMID 20109030.
- ↑ World Health Organization (June 2018). "BCG vaccine: WHO position paper, February 2018 – Recommendations". Vaccine 36 (24): 3408–3410. doi:10.1016/j.vaccine.2018.03.009. ISSN 0264-410X. PMID 29609965.
- ↑ "Leprosy post-exposure prophylaxis: innovation and precision public health". The Lancet Global Health 9 (1): e8–e9. 2021-01-01. doi:10.1016/S2214-109X(20)30512-X. ISSN 2214-109X. https://www.sciencedirect.com/science/article/pii/S2214109X2030512X.
- ↑ Yamazaki-Nakashimada, Marco Antonio; Unzueta, Alberto; Berenise Gámez-González, Luisa; González-Saldaña, Napoleón; Sorensen, Ricardo U. (2020-08-02). "BCG: a vaccine with multiple faces". Human Vaccines & Immunotherapeutics 16 (8): 1841–1850. doi:10.1080/21645515.2019.1706930. ISSN 2164-554X. PMID 31995448. PMC 7482865. https://pubmed.ncbi.nlm.nih.gov/31995448.
- ↑ "Leprosy Vaccine". American Leprosy Missions. Archived from the original on November 15, 2015. Retrieved October 20, 2015.
- ↑ "Trial set for world's first leprosy vaccine". The Guardian. June 6, 2014. Archived from the original on October 11, 2015. Retrieved October 20, 2015.
- ↑ "China's Mars plans, leprosy vaccine and self-driving taxis". Nature 537 (7618): 12–13. 2016-08-31. doi:10.1038/537012a. PMID 27582199.
- ↑ "WHO | MDT and drug resistance". WHO. Retrieved 2019-07-22.
- ↑ Reibel, F.; Cambau, E.; Aubry, A. (2015-09-01). "Update on the epidemiology, diagnosis, and treatment of leprosy". Médecine et Maladies Infectieuses 45 (9): 383–393. doi:10.1016/j.medmal.2015.09.002. ISSN 0399-077X. PMID 26428602.
- ↑ "Leprosy". www.who.int. Retrieved 2021-02-06.
- ↑ 87.0 87.1 87.2 87.3 Somar, Pmw; Waltz, M. M.; van Brakel, W. H. (2020). "The impact of leprosy on the mental wellbeing of leprosy-affected persons and their family members - a systematic review". Global Mental Health (Cambridge, England) 7: e15. doi:10.1017/gmh.2020.3. ISSN 2054-4251. PMID 32742673. PMC 7379324. https://pubmed.ncbi.nlm.nih.gov/32742673.
- ↑ Rao, Deepa; Elshafei, Ahmed; Nguyen, Minh; Hatzenbuehler, Mark L.; Frey, Sarah; Go, Vivian F. (02 15, 2019). "A systematic review of multi-level stigma interventions: state of the science and future directions". BMC medicine 17 (1): 41. doi:10.1186/s12916-018-1244-y. ISSN 1741-7015. PMID 30770756. PMC 6377735. https://pubmed.ncbi.nlm.nih.gov/30770756.
- ↑ "World Health Organization- Weekly Epidemiological Record. Geographical distribution of new leprosy cases, 2016".
- ↑ "Leprosy new case detection rates, 2016". WHO. WHO. Retrieved 19 December 2019.
- ↑ "Mortality and Burden of Disease Estimates for WHO Member States in 2002" (xls). World Health Organization. 2002. Archived from the original on 2013-01-16.
- ↑ "WHO | Leprosy: new data show steady decline in new cases". WHO. Retrieved 26 February 2020.
- ↑ 93.0 93.1 93.2 "WHO | Global leprosy update, 2015: time for action, accountability and inclusion". WHO. Retrieved 2019-01-14.
- ↑ Maggie Veatch (21 Feb 2019). "Leprosy still lurks in United States, study says". CNN.
- ↑ "About ILEP". ILEP. Archived from the original on 2014-08-12. Retrieved 2014-08-25.
- ↑ admin (2016-02-11). "Epidemiology of Leprosy". International Textbook of Leprosy. Retrieved 2019-07-23.
- ↑ 97.0 97.1 97.2 World Health Organization. (1985). "Epidemiology of leprosy in relation to control. Report of a WHO Study Group". World Health Organ Tech Rep Ser (Geneva: World Health Organization) 716: 1–60. ISBN 978-92-4-120716-4. OCLC 12095109. PMID 3925646.
- ↑ 98.0 98.1 98.2 "WHO | The Global Leprosy Strategy". WHO. Retrieved 2021-02-09.
- ↑ "WHO | Elimination of leprosy FAQ". WHO. Retrieved 2021-02-09.
- ↑ 100.0 100.1 "Leprosy elimination: World Health Organization". Archived from the original on 2014-03-14. Retrieved 2019-07-03.