Pluripotent stem cells
Welcome to the Wikiversity learning project for pluripotent stem cells. This project provides learning resources that help participants learn about stem cells and efforts to produce useful stem cells for medical therapies. Participants should feel free to ask questions and explore related topics.
For many patients, donated organs and transplanted tissues are used as replacements for body parts that are damaged or not functioning correctly due to a disease. Pluripotent stem cells are the subject of on-going research as a potential source of replacement cells and tissues for medical treatments. The potential medical uses of pluripotent stem cells are largely a matter of speculation, but it has been suggested that stem cell research might eventually lead to treatments for neurodegenerative diseases such as Parkinson's and Alzheimer's and allow for some recovery of neurological functions following spinal cord injury and stroke. Pluripotent stem cell methods might lead to improved treatments for tissues damaged by burns, heart disease, diabetes, osteoarthritis and rheumatoid arthritis.
Cells
[edit | edit source]Cells in the human body are fundamental functional and structural components. Most cells in your body are specialized for particular functions such as muscle cells that provide efficient contraction and allow for movement of your body.
Stem cells
[edit | edit source]In many tissues there are "adult stem cells" that have not yet fully specialized. Naturally existing "adult" stem cells provide a limited capacity for your body to replace old and damaged cells.[1]
A goal of medical research is to learn how to control and make use of stem cells to allow for new and improved medical intervention to treat tissue damage. Stem cells are usually restricted in terms of their developmental potential. Left to themselves, most adult stem cells differentiate in one of a few related cell types. However, during embryonic development, there are some stem cells that have the capacity to differentiate into all cell types: they are "pluripotent".
Note: There is interest in the idea that it might be possible to isolate pluripotent stem cells from adults. For example, see: Bone-marrow-derived stem cells - our key to longevity?
Discussion
[edit | edit source]What happens to the number of adult stem cells in human tissues as we grow older?
Pluripotent cells
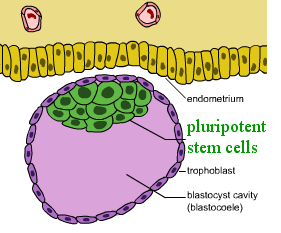
[edit | edit source]Pluripotent stem cells are normally very rare and naturally exist within early embryos as shown in Figure 1. Since 1981, methods have been available to isolate and grow embryonic stem cells in laboratories.[2]
Similar methods have been available since the late 1990s for human embryo-derived pluripotent stem cells,[3] but stem cell therapies cannot use just any stem cells. Stem cells should be derived from each patient in order to prevent rejection of the cells.

Reprogramming adult cells
[edit | edit source]In order to create pluripotent stem cells for patients that will not be immunologically rejected, nuclear transfer has been extensively studied. As shown in Figure 2, nuclei can be removed from differentiated cells of a patient and injected into oocytes. The cytoplasm of oocytes contains proteins that can reprogram nuclear DNA to an embryonic stem cell state.
Another approach to reprogramming of adult cells is to make use of proteins from cultured embryonic stem cells[4]. Simple fusion of somatic cells and embryonic stem cells results in tetraploid cells in which the DNA from the somatic cells seems to be reprogrammed to a stem cell state.
The DNA of specific genes important for the embryonic stem cell state such as Oct4 have been shown to be reprogrammed following somatic cell-embryonic stem cell fusion[5]. Such DNA reprogramming involves modifications such as methylation.
Discussion
[edit | edit source]Reading: Responsible Oversight of Human Stem Cell Research: The California Institute for Regenerative Medicine's Medical and Ethical Standards by Geoffrey P. Lomax, Zach W. Hall and Bernard Lo in PLoS Medicine (2007) 4(5): e114.
Q. Why do some governments restrict funding for stem cell research while others do not?
What makes a stem cell behave like a stem cell?
[edit | edit source]Each specialized cell type actively transcribes a subset of all the available genes. Key regulatory proteins that control which genes are active in any given cell type are DNA binding transcription control proteins.
The possibility exists that embryonic stem cells are normally created by making sure that oocytes contain the correct proteins and mRNAs needed to initiate the embryonic stem cell pattern of transcription. Potentially, all of the required proteins for specifying the embryonic stem cell fate might be DNA-binding transcription factors and associated regulatory proteins. Several transcription factors have been found to be particularly important for establishing and maintaining embryonic stem cells.
Oct4
[edit | edit source]
One of the key proteins involved in reprogramming adult cell nuclei to a stem cell state is the transcription factor Oct4. Working with mice as a convenient laboratory mammal, it was found in 1990 that Oct4 is stored inside oocytes and expressed at high levels in in embryonic stem cells[6]. Daughter cells of the embryonic stem cells that begin to specialize and differentiate during mouse embyogenesis were reported to reduce their levels of Oct4 protein[7].
The functional roles of Oct4 inside embryonic stem cells has been intensively studied in mice. Figure 5 shows a summary of many known actions of Oct4 in mouse stem cells. [8] In this set of experiments, 1155 gene transcripts were identified as being expressed in a similar pattern to that of Oct4; all but 69 of these transcripts were positively correlated with Oct4. Expression of Nanog and Sox2 (see below for more on these embryonic stem cell transcription factors) were correlated to Oct4 expression at 97% and 49% in these mouse experiments.

Sox2
[edit | edit source]Oct4 and Sox2 are thought to cooperate in binding to regulatory sequences of some developmentally important genes[10]. 392 of the genes with expression correlated to Oct4 that were identified by Rudnicki et al[8] were shown to have possible gene regulatory sequences for coordinately binding Oct4 and Sox2. Figure 4 shows Oct and Sox transcription factor proteins bound to DNA.
Nanog
[edit | edit source]Mouse embryonic stem cell growth in cell culture is generally dependent on a growth factor called leukemia inhibitory factor (LIF). In 2003, two research groups reported that Nanog is a transcription factor that allows mouse embryonic stem cells to grow in culture independent of LIF[11][12]. The Nanog gene is positively regulated by Oct4 and Sox2[13]. The Nanog protein is thought to be able to cooperate with Oct4 and Sox2 in maintaining the embryonic stem cell state.
Human stem cells
[edit | edit source]As discussed above, most research on embryonic stem cells has been performed with mice. Many similarities have been found in the behavior of human embryonic stem cells, but there are some differences from mice.
Human embryonic stem cells do not require LIF for growth in culture, but they express Nanog[14]. Artificially reducing the level of Nanog protein in human embryonic stems cells by means of RNAi knockdown results in differentiation of the cells[14][15].
Much less is known about embryonic stem cell proteins such as Oct4 and how they function in humans than in mice, but human Oct4 is also expressed in human embryonic stem cells [16]. In humans, Oct4 is expressed in several adult cell types, so it is not the case that cells with Oct4 are stem cells while non-stem cells fail to produce Oct4. That Oct4 functions to maintain human embryonic stem cells in an undifferentiated state is indicated by experiments in which RNA interference was used to inhibit Oct4 expression resulting in differentiation of human embryonic stem cells[17][18].
Gene transfer and stem cell induction
[edit | edit source]The results discussed above suggested the possibility that it might be possible to reprogram adult cells to take on the embryonic stem cell phenotype by forcing them to express a few key regulatory proteins such as Oct4 and Sox2.
Oct4, Sox2, Nanog and one additional protein, LIN28 (another embryonic stem cell-associated transcription factor [19]), have been used to transform somatic cells into human embryonic stem cells[20]. Retroviral vectors were used to insert the Oct4, Sox2, Nanog and LIN28 genes into target cells[21]. Oct4 and Sox2 were the key genes. Nanog, and to a lesser extent, LIN28, enhanced the recovery of embryonic stem cell-like clones. The embryonic stem cell-like clones were called "iPS" cells (Induced Pluripotent Stem Cells) and further investigations are needed to determine if adult somatic cells can be converted to iPS cells using Oct4 and Sox2 and if such cells can be efficiently induced to differentiate into the desired types of cells for medical uses.
Similar results were reported for Oct4, Sox2, Klf4, and c-Myc[22]. A major problem with using the protoncogene c-Myc is that it can cause tumors. Both research groups[20],[22] used retroviruses to insert the Oct4 and Sox2 genes into somatic cells. Other possible methods might be safer for medical uses.

.
Control over fate of embryonic stem cells
[edit | edit source]Sox2 is one member of the Sox transcription factor family[23]. There is interest in the idea that many cell fate changes during embryonic development involve switches in which Sox family transcription factors are expressed in differentiating cells. For example, Sox17 [24] has been suggested to function in developing endoderm to produce a control signal for cardiac myogenesis in nearby primitive mesoderm. This suggests that there might be [[w:Growth factor|growth factors] that can be added to cultured embryonic stem cells that would cause them to differentiate into specific types of cells such as cardiomyocytes.
Based on results obtained by using mouse embryonic stem cells, it has been suggested that a mixture of growth factors (TGF-ß1, BMP-2 and BMP-4, activin-A, VEGF-A, IL-6, FGF-2 and -4, IGF-1 and -2, and EGF efficiently converts the stem cells to cardiac progenitor cells that can be engrafted into heart tissue[25]. Similar results have been reported for human embryonic stem cells treated with activin-A and BMP-4[26] and the resulting cardiac mucle cells have been shown to have beneficial effects upon transfer to damaged rat hearts.
References
[edit | edit source]- ↑ Skeletal muscle stem cells by Jennifer C. J. Chen and David J. Goldhamer in Reprod Biol Endocrinol (2003) 1: 101.
- ↑ Establishment in culture of pluripotential cells from mouse embryos by M. J. Evans and M. H. Kaufman in Nature (1981) Volume 292 pages 154-156.
- ↑ Embryonic stem cell lines derived from human blastocysts by James A. Thomson, Joseph Itskovitz-Eldor, Sander S. Shapiro, Michelle A. Waknitz, Jennifer J. Swiergiel, Vivienne S. Marshall and Jeffrey M. Jones in Science (1998) Volume 282 pages 1145-1147.
- ↑ Nuclear reprogramming of somatic cells after fusion with human embryonic stem cells by C.A. Cowan, J. Atienza, D.A. Melton and K. Eggan in Science (2005) Volume 309 pages 1369-1373.
- ↑ Epigenetic Reprogramming of OCT4 and NANOG Regulatory Regions by Embryonal Carcinoma Cell Extract by Christel T. Freberg, John Arne Dahl, Sanna Timoskainen, and Philippe Collas in Molecular Biology of the Cell (2007) Volume 18 pages 1543–1553.
- ↑ New type of POU domain in germ line-specific protein Oct-4 by H.R. Schöler, S. Ruppert, N. Suzuki, K. Chowdhury and P. Gruss in Nature (1990) Volume 344 page 435-439.
- ↑ A POU-domain transcription factor in early stem cells and germ cells of the mammalian embryo by M.H. Rosner, M.A. Vigano, K. Ozato, P.M. Timmons, F. Poirier, P.W. Rigby and L.M. Staudt in Nature (1990) Volume 345 pages 686-692.
- ↑ 8.0 8.1 8.2 Oct4 Targets Regulatory Nodes to Modulate Stem Cell Function by Pearl A. Campbell, Carolina Perez-Iratxeta, Miguel A. Andrade-Navarro and Michael A. Rudnicki in PLoS ONE (2007) 2(6): e553.
- ↑ Molecular Basis for Synergistic Transcriptional Activation by Oct1 and Sox2 Revealed from the Solution Structure of the 42-kDa Oct1·Sox2·Hoxb1-DNA Ternary Transcription Factor Complex by David C. Williams, Jr., Mengli Cai and G. Marius Clore in Journal of Biological Chemistry (2003), Vol. 279, Issue 2, 1449-1457
- ↑ Crystal structure of a POU/HMG/DNA ternary complex suggests differential assembly of Oct4 and Sox2 on two enhancers by Attila Reményi, Katharina Lins, L. Johan Nissen, Rolland Reinbold, Hans R. Schöler and Matthias Wilmanns in Genes and Development (2003) Volume 17 pages 2048-2059
- ↑ The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells by K. Mitsui, Y. Tokuzawa, H. Itoh, K. Segawa, M. Murakami, K. Takahashi, M. Maruyama, M. Maeda and S. Yamanaka in Cell (2003) Volume 113 pages 631-642.
- ↑ Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells by I. Chambers, D. Colby, M. Robertson, J. Nichols, S. Lee, S. Tweedie and A. Smith in Cell (2003) Volume 113 pages 643-655.
- ↑ Transcriptional Regulation of Nanog by OCT4 and SOX2 by David J. Rodda, Joon-Lin Chew, Leng-Hiong Lim, Yuin-Han Loh, Bei Wang, Huck-Hui Ng and Paul Robson in Journal of Biological Chemistry (2005) Volume 280 pages 24731-24737.
- ↑ 14.0 14.1 Downregulation of NANOG Induces Differentiation of Human Embryonic Stem Cells to Extraembryonic Lineages by Louise Hyslopa, Miodrag Stojkovica, Lyle Armstronga, Theresia Waltera, Petra Stojkovica, Stefan Przyborskic, Mary Herberta, Alison Murdocha, Tom Strachana and Majlinda Lako in Stem Cells (2005) Volume 23 pages 1035-1043.
- ↑ Differentiation of Mouse Embryonic Stem Cells after RNA Interference-Mediated Silencing of OCT4 and Nanog by Shelley R. Hougha, Ian Clementsb, Peter J. Welcha and Kristin A. Wiederholt in Stem Cells (2006) Volume 24 pages 1467-1475.
- ↑ Oct-4 mRNA and protein expression during human preimplantation development in Molecular human reproduction (2005) Volume 11 pages 173-181.
- ↑ Specific Knockdown of Oct4 and ß2-microglobulin Expression by RNA Interference in Human Embryonic Stem Cells and Embryonic Carcinoma Cells by Maryam M. Matina, James R. Walsha, Paul J. Gokhalea, Jonathan S. Drapera, Ahmad R. Bahramia, Ian Mortona, Harry D. Moorea and Peter W. Andrews in Stem Cells (2004) 22:659-668.
- ↑ Oct-4 Knockdown Induces Similar Patterns of Endoderm and Trophoblast Differentiation Markers in Human and Mouse Embryonic Stem Cells by David C. Hay, Linda Sutherland, John Clark and Tom Burdon in Stem Cells (2004) 22:225-235.
- ↑ Assessing self-renewal and differentiation in hESC lines by Jingli Cai, Jia Chen, Ying Liu, Takumi Miura, Yongquan Luo, Jeanne F. Loring, William J Freed, Mahendra S Rao and Xianmin Zeng in Stem Cells (2006) 24(3): 516–530.
- ↑ 20.0 20.1 Induced Pluripotent Stem Cell Lines Derived from Human Somatic Cells by Junying Yu, Maxim A. Vodyanik, Kim Smuga-Otto, Jessica Antosiewicz-Bourget, Jennifer L Frane, Shulan Tian, Jeff Nie, Gudrun A. Jonsdottir, Victor Ruotti, Ron Stewart, Igor I. Slukvin, James A. Thomson in Science (2007) November 20; published online.
- ↑ Lentiviral vectors for enhanced gene expression in human hematopoietic cells by Ali Ramezani, Teresa S. Hawley and Robert G. Hawley in Molecular Therapy (2000) 2(5):458-469.
- ↑ 22.0 22.1 Induction of pluripotent stem cells from adult human fibroblasts by defined factors by K. Takahashi, K. Tanabe, M. Ohnuki, M. Narita, T. Ichisaka, K. Tomoda and S. Yamanaka in Cell (2007) Nov 30;131(5):861-72.
- ↑ Control of Cell Fate and Differentiation by Sry-related High-mobility-group Box (Sox) Transcription Factors by Véronique Lefebvre, Bogdan Dumitriu, Alfredo Penzo-Méndez, Yu Han and Bhattaram Pallavi in Int J Biochem Cell Biology (2007) 39(12): 2195–2214.
- ↑ Sox17 is essential for the specification of cardiac mesoderm in embryonic stem cells by Yu Liu, Masanori Asakura, Hironori Inoue, Teruya Nakamura, Motoaki Sano, Zhiyv Niu, Michelle Chen, Robert J. Schwartz and Michael D. Schneider in Proc Natl Acad Sci U S A (2007) Volume 104 pages 3859–3864.
- ↑ Cardiopoietic programming of embryonic stem cells for tumor-free heart repair by Atta Behfar, Carmen Perez-Terzic, Randolph S. Faustino, D. Kent Arrell, Denice M. Hodgson, Satsuki Yamada, Michel Puceat, Nicolas Niederländer, Alexey E Alekseev, Leonid V. Zingman and Andre Terzic in The Journal of Experimental Medicine (2007) Volume 204 pages 405-420.
- ↑ Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts by M.A. Laflamme, K.Y. Chen, A.V. Naumova, V. Muskheli, J.A. Fugate, S.K. Dupras, H. Reinecke, C. Xu, M. Hassanipour, S. Police, C. O'Sullivan, L. Collins, Y. Chen, E. Minami, E.A. Gill, S. Ueno, C. Yuan, J. Gold and C.E. Murry in Nature Biotechnology (2007) Volume 25 pages 1015-1024.
Additional reading
[edit | edit source]- From the USA National Institutes of Health website: Frequentlt Asked Questions Page for stem cells.
- Stem Cells and the Future of Regenerative Medicine (2002) Report from The National Research Council and the Institute of Medicine Committee on the Biological and Biomedical Applications of Stem Cell Research on the potential of stem cell research in the development of medical therapies and basic biological knowledge.
Additional resources
[edit | edit source]- National Public Radio report (audio available): Scientists Create Embryonic Stem Cells from Skin by Joe Palca. November 20, 2007.
