High School Chemistry/Atomic Structure, The Periodic Table, and Electrons or Light
- Background Knowledge
Observation vs. Inference: An observation is what is present while an inference is a guess based on the observation (the men are wearing coats vs. it is winter [because the men are wearing coats]).
History
[edit | edit source]- 440 BC - Democritus - First to propose that matter was made up of invisible and indivisible particles called atomos. His model consisted of what he depicted as an "atom", such as a shaded circle with a diagonal line through it or a shaded diamond.
- 1700s - Scientists all over the world using newly improved balances came up with three basic laws.
- Three Basic Laws
- Lavoisier proposed Law of Conservation of Mass--matter is neither created or destroyed during chemical reactions.
- Proust proposed Law of Constant Composition/Definite Proportions--composition of compounds/molecules is always the same, regardless of how the substance was made or where the substance is found.
- Law of Multiple Proportions states that elements within compounds/molecules always combine in whole number ratios.
3. 1803 - John Dalton's atomic theory:
- All matter is made up of atoms
- All atoms of the same element are identical and unique. (WRONG! All atoms of a given element are NOT identical due to isotopes)
- Atoms can't be created, subdivided or destroyed. (WRONG! Atoms can be broken down into protons, neutrons and electrons)
- In chemical reactions, atoms can only be combined, separated or rearranged; atoms of 1 element can't be changed into atoms of another element. (WRONG! Atoms can be changed into other atoms (in nuclear reactions)).
John Dalton created the solid sphere model.
4. 1897/1898 - J. J. Thomson passed an electric current through a glass tube (vacuum). He noticed a glow, which he termed it the "cathode ray" (electrons moving from positive to negative). He discovered that cathode ray particles are negatively charged and are extremely tiny (electrons are 1,000 times smaller than the atoms that belong to hydrogen).

J. J. Thomson came up with the Plum pudding model, which depicts electrons embedded in a loose mass of protons (nucleus had not yet been discovered) like raisins in plum pudding.
5. 1908/1913 - Robert Millikan calculated the exact mass and charge of electrons; performed the Oil drop experiment: Electrons are massless compared to protons and neutrons. Two inferences can be made from the given electron knowledge: Because atoms are electrically neutral, they must have a positive charge to balance out the negatively charged electrons. Because electrons have so much less mass than atoms, atoms must contain other particles to account for their mass.
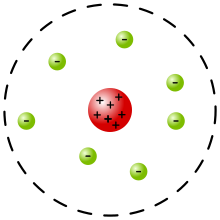
6. 1909/1910 - Ernest Rutherford found out about the nucleus by performing the Gold Foil Experiment. Rutherford shot alpha particles (very small, positively charged helium nuclei) through a sheet of gold foil, expecting that all of the alpha particles would pass straight through gold foil. This did not happen--a few of the alpha particles were actually deflected/redirected. Rutherford concluded that atoms must have a very dense part and a very spacious part. The dense part must be very tiny because so few alpha particles were redirected; the small area must be positively charged because it reflected positively charged alpha particles. Rutherford discovered the nucleus (super tiny/positively charged or dense). Rutherford's Nuclear Model included a nucleus (positively charged) in the center of the atom, surrounded by a loose cloud of dense electrons and most of the atom is empty space.
7. 1922 - Niels Bohr discovered that each orbit [of electrons] has a specific energy level like planets around the sun. The energy of electrons increases as you move away from the nucleus. Electrons must absorb energy to jump to outer levels, and they must release energy when falling back to lower energy levels (in the form of electromagnetic radiation). Examples are visible light, UV, infrared, etc. Bohr came up with the Planetary Model.
8. 1926 - Erwin Schrodinger discovered the Quantum Mechanical Model. He gave the mathematical description of the location of electrons.
9. 1932 - Sir James Chadwick discovered the neutrons. He also found out that all atoms past helium are heavier than just protons, therefore, atoms must contain another particle with a neutral charge, but with approximately same mass as the protons.
10. 1964 - Murray Gell-Mann and George Zweig discovered quarks (subatomic particles). See also the Quark Model: w:Quark model.
11. 2015 - Large Hadron Collider: Higgs boson.