Formulas, equations, and reactions
| Subject classification: this is a chemistry resource. |
| Type classification: this is a lesson resource. |
| Completion status: Ready for testing by learners and teachers. Please begin! |
Formulas
[edit | edit source]In studying chemistry, we represent the composition of a substance through the use of the chemical formula, a symbolic representation of a chemical reaction. The chemical formula of a given substance, represented by either a single uppercase letter or an uppercase letter followed by a lowercase letter, specifies the elements that the substance contains through the use of atomic symbols, and is sometimes marked with numerical subscripts that show the relative proportion of each element to the other elements in the substance.
Molecular formula
[edit | edit source]There are currently 118 elements listed in the Periodic Table, from which molecules can be made. The molecular formula simply lists every element, and the number of atoms of each element, in a molecule or compound.
Examples
[edit | edit source]
Let's use the formula for magnesium chloride, MgCl2. If we interpret this, it shows us that for every one magnesium atom in the compound, there are two chlorine atoms (thus the subscript "2" attached to the Cl).
Now consider the formula for carbon tetrachloride, CCl4. Once again, we interpret this as there being one carbon atom for every four chlorine atoms.
Structural formula
[edit | edit source]The structural formula follows the methods of a standard chemical formula, however it also denotes a structural representation of a chemical reaction. It indicates, with detail, the arrangement of atoms in a molecule or compound. Chemical structure is out of the scope of this section and level.
Examples
[edit | edit source]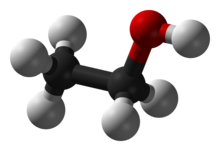
Ethanol has the molecular formula C2H6O. However, its structure involves an -OH group, so the molecular formula does not fully represent this structure. Therefore, the structural formula could be represented by C2H5OH. You could also go into further detail and show the full structural formula: CH3CH2OH.
The amount of detail you go to really depends on whether the molecular formula could be misconstrued as something else. For example, both propanone and propene-2-ol have the molecular formula C3H6O, but their structures are quite different. Propanone is best represented by CH3C(O)CH3, while propene-2-ol is best represented by CH2C(OH)CH3. This is why the structural formula is so important - you can't always tell what a substance is just from its molecular formula
Empirical formula
[edit | edit source]The Empricial formula is the simplest whole number ratio of atoms in a compound. Let us take C6H16 for example. Notice although the compound is in whole number ratios, it is not in the simplest form. Thus we can divide the whole numbers by 2, to yield C3H8 which is now the simplest whole number ratio of C6H16. Thus makes C3H8 an empirical formula and C6H16 a molecular formula.
But now we need to look at the two main types of substances: ionic and molecular.
Ionic Substances and Ionic Nomenclature
[edit | edit source]For more information, see Ionic bond
An ion is defined as an electrically charged particle that has acquired an overall (net) charge by losing or gaining electrons. One of the most familiar examples of an ionic substance (a substance composed of ions) is sodium chloride, NaCl, commonly known as "table salt".
Let's take copper for example: copper is normally found in a neutral state, meaning there are no extraneous electrons hanging around, nor does it need to gain any electrons to become neutral. However, in a reaction with hydrochloric acid, HCl, copper will normally lose an electron to chlorine when bonding to it, and become CuCl2, or copper(II) chloride.
In this process, copper has become a cation. A cation is an atom that has lost one or more electron(s) to become positive. We would denote this by showing coppers atomic symbol with a superscript denoting it's charge - Cu2+. This symbol shows that copper has lost two electrons to become positively charged. Other atoms, such as sodium in the case with sodium chloride, become Na+, denoting that it has lost one electron.
Yet in the same process with CuCl2, chlorine has gained one electron, making it an anion. An anion is an atom that has gained an extra electron to become negative.. This we denote with the atomic symbol for chlorine, and place a superscript "1-" or just "-", shown as Cl- or Cl1-.
So now it only seems logical that when you place cations and ions together (that will presumably react), you get what is called an ionic compound. You've already been shown two such compounds: NaCl and CuCl2.
Now let's discuss the formula unit. The formula unit is the group of atoms explicitly present in the compound. For example, NaCl denotes one Na atom for every Cl atom, nothing more, nothing less. The same goes with CaCl2, which denotes one Ca atom for every two Cl atoms. A formula unit is the smallest unit for each of these substances.
But how do we write these formulas? Simple enough. Take the reaction between Fe3+ and SO42-. When you place them together - assuming that they will, in fact, react - we take the superscripts and place them as subscripts. Here's a more visual example:
References
[edit | edit source]- Flowers, Paul, Klaus Theopold, Richard Langley, William R. Robinson, Mark Blaser, Simon Bott, Donald Carpenetti, Andrew Eklund, Emad El-Giar, Don Frantz, Paul Hooker, George Kaminski, Jennifer Look, Carol Martinez, Troy Milliken, Vicki Moravec, Jason D. Powell, Thomas Sorensen, and Allison Soult. Chemistry. N.p.: n.p., 2015. Chemistry. OpenStax College, Mar. 2015. Web.
