Prebiotic chirality
| Completion status: this resource has reached a high level of completion. |
| Subject classification: this is a biology resource. |
| Type classification: this is an article resource. |
| Educational level: this is a research resource. |
The mechanical and geometrical origin of chirality and the homochirality of glycerol-phosphate, glyceraldehyde-phosphate and prebiotic amino acids and major physico-chemical characteristics of these amino acids.
abstract
Bringing closer phospholipids each other on a bilayer of liposome, causes their rotation around their fatty acids axis, generating a force which brings closer the two sheets of the bilayer. In this theoretical study I show that for getting the greater cohesion of the liposome, by these forces, the serine in the hydrophilic head must have a L chirality. In the case where the hydrophilic head is absent amino acids with L chirality could contribute to this cohesion by taking the place of L-serine. Some coenzymes having a configuration similar to ethanolamine may also contribute. This is the case of pyridoxamine, thiamine and tetrahydrofolic acid.
The grouping of amino acids of L chirality and pyridoxamine on the wall could initialize the prebiotic metabolism of these L amino acids only. This would explain the origin of the homo-chirality of amino acids in living world.
Furthermore I show that in the hydrophilic head, the esterification of glycerol-phosphate by two fatty acids go through the positioning of dihydroxyacetone-phosphate and L-glyceraldehyde-3-phosphate, but not of D-glyceraldehyde-3-phosphate, prior their hydrogenation to glycerol-3- phosphate. The accumulation of D-glyceraldehyde-3-phosphate in the cytoplasm displace the thermodynamic equilibria towards the synthesis of D-dATP from D-glyceraldehyde-3-phosphate, acetaldehyde and prebiotic adenine, a reaction which does not require a coenzyme in the biotic metabolism. D-dATP and thiamine, more prebiotic metabolism of L-amino acids on the wall, would initialize D-pentoses phosphate and D-nucleotides pathways from the reaction of D-glyceraldehyde-3-phosphate + dihydroxyacetone-phosphate + prebiotic nucleic bases.
The exhaustion of the prebiotic glyceraldehyde (racemic) and the nascent biotic metabolism dominated by D-glyceraldehyde-3-phosphate, would explain the origin of homo-chirality of sugars in living world.
https://en.wikiversity.org/wiki/Prebiotic_Petroleum
https://en.wikiversity.org/wiki/Prebiotic_chemo-osmosis
https://en.wikiversity.org/wiki/Prebiotic_chirality.
français
Note on 14.03.2015: This article is part of the summary of my work until 2014, published in Origins of Life and Evolution of Biospheres, March 2015.
Reference: Prebiotic Petroleum; Mekki-Berrada Ali, Origins of Life and Evolution of Biospheres, 2015, DOI 10.1007/s11084-015-9416-7.[1]
Introduction
[edit | edit source]
In what follows I have shortened the terms below because they are widely used:
amino acid: aa
phospholipide: PLD
phosphate: P.
This theoretical study is made under the hypothesis of a prebiotic molecular evolution that would occur in a pocket of abiotic oil of geochemical origin ( pétrole prébiotique, being prepared). The theoretical development of this hypothesis and literature has shown that it is entirely possible that liposomes can be formed under these conditions from aqueous vesicles present in the oil phase that migrate into the water phase to form these liposomes. The wall of the vesicles would formed by the head of carboxylic fatty acids and the water content of these vesicles, and the main phase water too, would contain small hydrophilic molecules which glycerol, ethanolamine, phosphate and certain racemic amino acids , as serine, for synthesize the hydrophilic head that we know by esterification.
That is trying to imagine the arrangement of these small molecules in hydrophilic head (in the vesicle or liposome) and the fixation of the head on the fatty acids, it occurred to me that this process can impose glycerol-P and aa chirality, and the sequestration of these aa on the wall, according to their chirality, but also of their length, their volume and their chemical function carried by their radical.
At first we study concatenation of PLD molecules, due to ethanolamine or choline or serine, which are part of the hydrophilic head. We will show that this arrangement by concatenation is the source of serine chirality. We then study the chirality of the glycerol-P whose origin is to find in a compromise between the steric hindrance and electronic repulsion between the P and the closest fatty acid.
A special chapter is dedicated to the sequestration of free aa because they will put in place of the hydrophilic heads, through their amine at the layout in the vesicles or at the loss of these heads in response to the outside environment.
We finally conclude by the properties of the homochirality of the liposomal wall which can cause the initialization of prebiotic metabolism in the same bilayer, assumption made in the work on prebiotic chemo-osmosis. The homochirality of biotic metabolism would be originated from that of the prebiotic bilayer.
Prebiotic serine chirality.
[edit | edit source] Serine, ethanolamine, choline and glycerol are attached to P through an ester bond. This part of the hydrophilic head is a movable arm. The second part of the hydrophilic head is the fixed arm, consisting of glycerol-P linked to 2 fatty acids. Even when the PLD molecule is isolated from other molecules of PLD, the fixed arm has high inertia. And when two PLD molecules are bringing closer each other by force of the hydrogen bond (NH2) or ionic bond (NH3+), to the two free oxygens of P, and by force of the hydrophobicity (displacement of 4 fatty acids), is the free arm to move first. This is all the faster and stronger than hydrogen bonding (or ionic bonding) is acting at a greater distance than do the Van der Waals forces of fatty acid hydrophobicity. This necessarily entails a rotation of the molecule PLD whole, around 2 fatty acids, because the amine NH2 (NH3+) is not aligned with the free arm, and the P to which it brings closer, is massive and very close to the fatty acids.
If we are positioned above the PLD with fatty acids below the plane defined by three oxygens of P, a counterclockwise rotation raise PLD moecule and clockwise rotation plunge it. Clockwise rotation therefore close the leaflet in question to the other leaflet of the bilayer, or to the oil phase for aqueous vesicle. The clockwise rotation is a very important factor of cohesion when taking into account the large number of PLD molecules that contains the bilayer. And it is this layout that will determine serine chirality and the position of the amine NH2 (NH3+) of ethanolamine.
Positioning ethanolamine NH2 in the phospholipid molecule to obtain the greatest possible liposome cohesion.
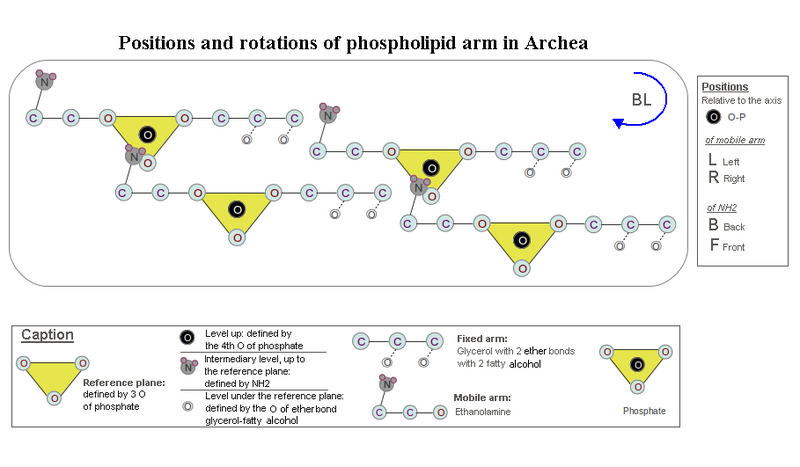
[edit | edit source]If you look toward the two free phosphate oxygens (see drawing 1), NH2 can occupy four possible positions: front-left (FL), left-back (BL), front-right (FR), right-back (BR). The BL is the only position to give a concatenation, with clockwise rotation, to a large number of PLD molecules.
Indeed, the 2 BR and FL positions rotate in the counterclockwise direction to reach the P. The other two positions rotate clockwise, but the FR position can put together only with a single other PLD, as NH2 and the two free oxygens of P are on the same side and neutralizes each other. On the other hand the position BL may concatenate PLD molecules while maintaining closer the two leaflets of the liposome by clockwise rotation.
-
Drawing 1. Hypothesis of the mechanical origin of chirality in the phospholipids. here is treated the positioning of ethanolamine relatively to the phosphate. Its displacement towards the phosphate to establish hydrogen bonding causes a rotation. This hypothesis states that the clockwise rotation and positioning in single file of the phospholipid molecules, (RG box), results in bringing closer the two leaflets of the liposome, increasing its cohesion. In this configuration the serine chirality would be L, because its carboxyl can not be put under the reference plane defined by the phosphate (see caption in the drawing) and then would be near the carboxyl of two fatty acid molecules neighbors.
The chirality of serine is L.
[edit | edit source]We have seen the mechanical origin of positionning the ethanolamine NH2. This also applies to the NH2 of serine. The carbon of the carboxylic function of serine positions by electron repulsion. In fact if you draw the layout of several PLD neighbors (in top view, see drawing 1-RG), we see that the PLD of a row fit together in the adjacent row and the carbon of the amine (NH2) is in the same level and aligned along an oblique line, with the carbons bearing the esters of glycerols.
If we position the carboxyl group of serine under the plane defined by P (which would give the D-serine), then it will find itself surrounded by four oxygen atoms of two ester bonds belonging to two molecules of successive PLD. There will then, in a confined space, six oxygens for a single hydrogen for hydrogen-bond, in the best cases depending on the pH. As the two carboxylic functions of the two fatty acids are fixed, following the overall coherence of the liposome, that of serine, mobile, is expelled by electron repulsion automatically above the plane defined by the P. Then we have L-serine.
In the case of archaea is no longer electron repulsion that occurs, but the steric hindrance. The carbon of the carboxylic function of serine would be inserted between the heads of fatty acids, which move the 2 PLD of the adjacent row, greatly increasing steric hindrance. But the steric hindrance should be minimal for these prokaryotes, as will see also for the chirality of glycerol-P of the fixed arm.
Prebiotic glycerol-phosphate chirality.
[edit | edit source] The chirality of the glycerol-P must be independent of the position of the movable arm as is the case between bacteria and archaea. These 2 domaines of procaryotes ensure the cohesion of the liposome with the same mechanical principle we have just seen, but the chiralities of the glycerol-P of the fixed arm are opposite. The chirality of the fixed arm depends on the binding of glycerol to aliphatic chains. Indeed the ester bond is large and push away electrons with its two oxygens, while the ether bond has only one oxygen and is less repulsive than each of the two oxygens of the ester bond (the ether linkage is known to be very stable, being little reactive).
In Archaea, with the ether bond of fatty acids to minimize the steric hindrance, the 2 ether bonds may be on the same side as the two free oxygen atoms of P, to fill the vacant space beneath the half-reference plane defined by P (see Drawing 2). As the position of the serine relative to P is imposed mechanically, as we saw above, the amine (NH2) and oxygen from the first ether bond are, at once, each in a different half-plane, the chirality of the glycerol-P is then levorotatory such as serine, since they are superimposed by rotation. Is an L-glycerol-3P if the terminal alcohol was above glycerol as the carbon of serine CO2H is above the membrane. Or to reduce steric hindrance, the terminal alcohol glycerol must be below its carbons. It is then a D-glycerol-3P or L-glycerol-1P, which is found in archaea.
With the ester bond, the electron repulsion between it and the free oxygens of P automatically positions aliphatic chain closest to the P on the same side as the amine of serine, on the other side of the half-plane defined by the P (see Drawing 1-RG). And by steric hindrance and repulsion with the first, second aliphatic chain is on the side of the half-plane containing the P and under the carbons of glycerol for the same reasons of steric hindrance for archaea. Hence the L-glycerol-3P (sn-glycerol-3P) of bacteria or D-glycerol-1P (see KEGG [2] for synonyms).
It is also noteworthy that in the vesicle or on the inner leaflet of the liposome, while the introduction of hydrophilic heads, fatty acids are glued side by side, the hydroxyl of one facing the oxygen of the other, the hydrogen bonding between them is reinforced. When attaching hydrophilic heads on a field of carboxylic carbons, half the job is done and only remain to position the head.
-
Drawing 2. See drawing 1 for caption.
Physico-chemical features of prebiotic amino acids.
[edit | edit source]Amino acids in the prebiotic chemio-osmose theory.
[edit | edit source] I have already studied the interaction aa-liposome in the theoretical paper on the prebiotic chemo-osmosis. I assumed as to form prebiotics pores, which, like the ionophores produced by some prokaryotes, contain amino acids (D and L) and alpha hydroxy acids (D and L), the aa had to intercalate between the P and the amine, with hydrogen bonds on the two sides. So position of their radical is vertically to the bilayer, inside or outside the bilayer.
The transort of aa across the bilayer, by prebiotic pores or unitarily, led me to suppose that the metabolism could start in the bilayer which then serves as a scaffold to position them next to each other. The effectiveness of these groups of aa would be growing if their homochirality increases more and more. But I had made no assumptions about the chirality, D or L, of the final aa.
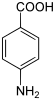
Similarly, the similarity of 13 biotic aa with ethanolamine and serine for the length of their carbon chain (2 C) between the head and the function carried by the radical, and the constraints they must undergo during their passage through the membrane, let me guess that prebiotic aa should be short and hydrophobic. On the other side the radicals very reactive or ionized were problematic for the passage through the membrane. These are the ionizable radicals: acids, amides, amines and those of the histidine and arginine.
Sequestration of aa in the theory of the mechanical cohesion of the liposome, that we will see just after, explains origin of their chirality L, origin that theory of prebiotic chemo-osmosis can not explain. However the two processes, diffusion through the membrane and sequestration of aa that we will see right away, are not exclusive of each other and can coexist simultaneously.
Amino acids sequestration in the mechanical cohesion of liposome theory.
[edit | edit source]Principle
[edit | edit source] We saw earlier that the chirality of the serine in the liposome has its origin in a force that brings closer the two leaflets of the liposome. It is a force perpendicular to the membrane. But it is only possible if the serine is attached to the P of the fixed arm by an ester bond. Besides any amine attached to P can serve this cohesion. This is the case of ethanolamine, choline but also glycerol with its terminal hydroxyl for hydrogen bonding.
However the mechanical cohesion of the liposome has two components, the component perpendicular to the liposome surface as we have seen and which I call vertical, but also a second component perpendicular to the plane defined by the two fatty acids of PLD and I will call horizontal stronger, and whose direction is from the amine to P of the following PLD. There is no cohesive force perpendicular to the horizontal force and contained in the plane of the fatty acids of the same PLD. This is what allows slippage of PLD chains against each other and differentiate the liposome from the rigidity of the crystal.
Horizontal cohesion has two parallel components, the first, the P-amine hydrogen bond, between two consecutive hydrophilic heads and the second consisting of the Van der Waals bonds between the aliphatic tails of 2 consecutive PLD. If these second component are the foundation of vesicle formation, since in vitro can be produced vesicles with fatty acids alone, the hydrogen bonds of the first component can be done and undone quickly locally, while maintaining the overall horizontal cohesion. Which compensates the inertia of the hydrophobic tails. In the vesicles without movable arm, with fatty acids alone, the carboxylic heads arranged head to tail can form hydrogen bonds, but do not form a global horizontal cohesion.
Also, if it happens that the ester bond of mobil arm, has to be hydrolyzed, or that any hydrophilic head comes running out, the place is free for any other molecule with an amine. This is what I call sequestration.
Sequestered amino acids chirality.
[edit | edit source]In the case of aa sequestered, the first component of horizontal cohesion is fully restored only if the NH2 (or even an OH) is accompanied by a P or a carboxylic carbon, as is the case of aa , to attach the amine of the PLD of the rear and which is at its level. These aa will be, of course, of L chirality as serine, since they are always in the same configuration, where there is no breach in the liposome (hole-free fatty acids) and the PLD chains will be side by side (see Drawing 1-RG above).
Others physico-chemical features of sequestered amino acids.
[edit | edit source]Space freed up by the hydrophilic head has a well-defined surface. That would be the geometric origin of these prebiotic aa. But their origin is also mechanical as they act mechanically as serine that is linked to P. So they have a mechanical and geometric origins.
- Horizontality: These aa are arranged horizontally in contrast to those who pass through the membrane, and wich arrangement could be random.
- The maximum length of these aa is approximately equivalent to 7 atoms (see Drawing 1 above), regardless of the carboxylic carbon. And the aa can not exceed this length, otherwise it would prevent the sliding of PLD chains neighbors and vice versa.
- The maximum width of the aa, or thickness, is limited to 4 carbons (see Drawing 1 above). This leads to small side chains as with val, thr, leu, ile, arg, or the thickness of a single aromatic ring as phe, trp, tyr, his.
- In height, outside the membrane, there is no limit.
The chemical functions carried by the radical of sequestered amino acids.
[edit | edit source]There is no question here of finding chains of chemical reactions of prebiotic metabolic network. But if sequestration by the mechanical cohesion favors some aa and excludes others, it would allow us to glimpse the initialization of prebiotic metabolism from some groups of aa.
- Carboxylyc acids and amides:
It is remarkable that there are two carboxyls functions and two amides functions at short chain, 1 and 2 atoms more than serine, in biotics aa! Why these lengths and dual function among the 20 biotics aa? The answer can it be from the mechanical cohesion? One can speculate that indeed, at these lengths, these two functions are at the level of P of the lost head and that they can attract the amine of the rear PLD by hydrogen bonding. This which would restore almost completely the first component of horizontal cohesion as like would do the P, while the carboxylic function of the head of aa, offset from the amine of the rear PLD, adds an imbalance in this restoration.
We can also assume, if the head keeps only the glycerol of the fixed arm, the free hydroxyl of the latter can establish a hydrogen bond with theses functions and even an ester bond with the carboxylic function. This which would restore the first horizontal component and the vertical component of the mechanical cohesion.
By against a carboxyl function or an amide function of a longer aa would reach the fatty acids, would be pushed away by electron repulsion and would destabilize the binding of aa.
- The radicals bearing a hydroxyl or an amine and arriving at the level of fatty acids, may make hydrogen bonds with the latter. This which partially restore the vertical component of cohesion. This is the case of arg and tyr (4 atoms than serine in length), lis and his (more than three atoms).
- The hydrophobic radicals, less reactives should fill the entire width of the freed space. This is a consolidation by steric hindrance. This is the case of 9 biotics aa that are either branched (leu, ile, val, thr) or aromatic (phe, trp, pro), or containing a sulfur atom (met, cys) that has as much influence sterically by its volume and its core that the phosphorus atom.
- There are only 2 biotics aa which only bring their heads to the mechanical cohesion: ala, gly.
- The problems raised by certain biotics aa such as met, cys, pro, trp:
Any atom bearing a radical of high action on its neighborhood should be excluded, such as transition metals, halogen, cations but also sulfur. In this case the Cys may have a non-mechanical origin and derived from the ester bond thiolysis of the P-serine, for example. Remains the origin of met. The pro can it establish a hydrogen bond in its cyclic form? Or the pro prebiotic she was linear? The trp should raise a problem height since no biotic aa has height! Does citrulline and ornithine would be sequestered as prebiotics aa?
From prebiotic to biotic homochirality.
[edit | edit source] We have seen the theoretical principles of the establishment of homochirality, assuming that required molecules are, either pre-existing amount in the reaction medium (prebiotic) such as fatty acids, or they can be easily recovered by shifting themodynamics equilibriums.
We shall study in this chapter the connection of these two processes taking into account the more stringent conditions of the hypothesis of prebiotic pocket oil and taking pathways of biotic metabolism the least complexes possible. And this to try to imagine the molecular evolution that could take place from prebiotic metabolisme to biotic metabolisme for this implementation of homochirality.
Prebiotic processes for the establishment of homochirality.
[edit | edit source] First come the fundamental processes of statistical thermodynamics in liquid (oil or water). The molecules diffuse freely in the medium, and interact with each other with energies increasing with temperature. In the organized environment of a cell, small molecules are not free and are controlled and even transported by macromolecules. The cytoplasm is considered as "colloidal gel".
It is this fundamental difference which forbids us to reproduce exactly the same enzymatic reactions of biotic metabolism with chemical reactions of a complex mixture, liquid, without macromolecules. The starting point of molecular evolution in our hypothesis of prebiotic pocket of oil, being the self-assembly of fatty acids synthesized abiotically, our goal we have introduced above, is to equate these two worlds ,to bring them closer , taking reactions of biotic metabolism closer to reactions of organic chemistry, and to consider the geochemical conditions of the pocket of oil closest to the physicochemical characteristics of living.
Conditions of prebiotic pocket oil.
[edit | edit source] The physico-chemical conditions closest to the characteristics of living are those, for example, bacteria living in the pockets of oil "fossil" at 55°C and 400-800 bar. [3]. But you can put out a little more, to go past slightly the maximum temperature of the multiplication of extremophiles from 113°C, to 150°C; pressure has less influence on the bacteria and can be doubled to 1.5 kbar as the Tupi oilfield offshore Brazil.
I think that under these conditions, the reactions that take place can be in thermodynamic equilibrium and thus continuously produce some molecules, if these ones are also trapped. This is the case of the hydroformylation that produce glyceraldehyde and dihydroxyacetone (100-140°C, 120 bar, from H2 CH2O CO; Györgydeák et al. 1998 [4]), the hydrogenation of glyceraldehyde to glycerol (50°C, 60 bar in the presence of H2) and the synthesis of aa and ethanolamine (A.D. Aubrey et al. (2009), Fig 5 [5]) and choline (analogous to synthesis of trimethylamine).
Assuming an abiotic oil formation by diagenesis (see article Prebiotic Petroleum ), where pressure and temperature rise gradually, CH2O H2 CO NH3, originally from clathrates (JL Charlou, [6] Ifremer serpentinization and synthesis inorganic hydrogen, methane and hydrocarbons along the Mid Atlantic), may exist and form glycerol, glyceraldehyde, dihydroxyacetone, ethanolamine, choline and few aa in small quantities.
Assuming a brutal production of abiotic oil (see article Prebiotic Petroleum ) by metamorphism during subduction or near these areas (accretion cones), the conditions of pressure and temperature are higher limits for the synthesis of oil by the FTT process. These conditions make possible the synthesis of glycerol and ethanolamine, directly as in industrial processes (500 °C and high pressures) from molecules of ethylene and propene originally from the FTT process. These high temperatures would not allow the formation of hydrophilic heads. should await the migration of the pockets of oil to areas with more moderate temperatures. At these temperatures the synthesis of these two molecules, ethylene and propene, no longer carried, it should be assumed that large amounts of glycerol and ethanolamine have been synthesized previously, along with fatty acids and in the same concentrations, to support the formation of hydrophilic heads. No results in the field of oil "fossil" or synthetic has so far been reported like this, for glycerol (fossil and synthetic oil, abscence of N2) or ethanolamine (fossil fuel, the presence N 2).
Establishment of prebiotic homochirality.
[edit | edit source] Let us therefore in the case of prebiotic pocket of oil formed slowly by diagenesis, where a network of chemical reactions are in thermodynamic equilibrium and can provide continuous P, glycerol, glyceraldehyde, dihydroxyacetone, ethanolamine, choline, serine and other aa. The concentrations of these molecules does not matter, because account only the possibility of trapping to shift equilibriums. And this trapping is done by fatty acids and PLD integrated into coherent macro-structures, difficult to hydrolyze, such as vesicles in the oil phase or liposomes in the water phase.
However for fixing the hydrophilic heads on the heads of fatty acid, all reactions are esterifications. Yet in the water hydrolysis tends to defeat them. And the oil phase, which would keep them, is reduced to the thickness of the bilayer, if this fixation was to be in bilayers of fatty acids of the water phase (equivalent liposomes without hydrophilic head).
- The vesicles of oil phase.
In the oil phase vesicles concentrate all hydrophilic molecules normally with little water. We saw in the prebiotic chemo-osmosis paper that the liposome in the water phase is the very foundation of life, since it defines a specific inside and a changing outside, that allows more and more organization inside by a communication (electronic and ionic) through the membrane. The vesicles of the oil phase, they can not communicate with the outside, the oil constituting an infinitely thick wall, allowing only the arrival of hydrophilic molecules. The vesicles are quickly isolated from each other.
Against by the vesicles of the oil phase have a true oil/water interface. In such an interface, the molecules of organic chemistry produced during the process of forming the prebiotic pocket of oil , can occupy the same two phases and would create an intermediate phase for molecules that are more or less soluble in the first 2 phases. This intermediate zone can be very thin, but it allows, with the two other phases, all possible reactions following the graduation of solvation phases. Particular it allows the esterification of fatty acids by the hydrophilic heads, hydrolysis of ester bonds becoming increasingly difficult with the extent of phospholipid surface.
- Synthesis and fixation by esterification of the hydrophilic head.
From what we know, the esterification is a reversible reaction. The direction of displacement of the equilibrium depends only on the solvent. For a given solvent steady-state does not depend on the temperature or pressure. Reactions are very slow in the abscence of catalyst. The catalyst is the proton H+ derived from HCl, H2SO4, H3PO4. Carboxylic heads are also catalysts (autocatalytic): see esters in wikipedia.
Also we will assume that esterifications that concern us are possible in the environment of the prebiotic pocket of oil and that they realize slowly, and even very slowly as geological time is not determined. However compared to the biotic metabolism, we will see in what follows, the role of the latter will act primarily on the number of intermediaries in networks of chemical reactions.
For example, the displacement of equilibrium of the racemic mixture of glycerol-P to glycerol-3P fixed by fatty acids, will necessitate removal of glycerol-1P or 2P of the surface of the fatty acids, and their hydrolysis and esterification of released glycerol to glycerol-3P, then diffusion of the latter to the surface of fatty acids. Fixing it then actually shift the equilibrium of glyceraldehyde to glycerol (hydrogenation reaction).
Given this large number of intermediates and the catalytic potential of the surface of fatty acids and / or hydrophilic heads, it is quite legitimate to consider the hydrogenation of racemic glyceraldehyde-P (or dihydroxyacetone) to glycerol-3P after attachment of the first on the fatty acid. Because the keto-enol tautomerism of the aldehyde function allows them to adopt the conformation imposed by the surface, as we have seen in the theoretical approach of the chirality of the liposome (Drawing 1-RG and Drawing 2). Once glyceraldehyde-P fixed and the conformation of glycéraldéhyde-3P performed then the hydrogenation, made possible by the presence of H2 (as we have suggested above in Conditions of prebiotic pocket oil) and by the catalytic influence of the surface, can be performed easily.
Among the living, the inertia generated by displacements of these many equilibria is eliminated through the transport of small molecules by macromolecules, and only the proper conformation is synthesized and protected from racemization by these macromolecules. Moreover it is easy to see that the mobility of some enzymes in the water phase (inside the liposome) is one of the first steps of molecular evolution because they can act sequentially to different locations on the surface, by taking the necessary small molecules with them. Otherwise, without this mobility, small molecules would set randomly. Then depending on the environment of the surface, catalysis could be more or less effective. And if an error occurs, the displacement of equilibriums should lengthen greatly the achievement.
- Formation of liposomes.
We saw earlier that the fatty acid bilayer vesicles in water not suitable for the formation of phospholipid molecules. Similarly the formation of heterogeneous liposomes, formed by random assembly of phospholipids and fatty acids, as described in the literature (eg Segré [7]), should lead difficultly, for the same reasons, to a homogeneous or structured liposome (hydrophilic heads with serine, choline or ethanolamine) with very little bare fatty acids.
The scenario for the formation of liposomes that I describe here, assumes that the conditions of temperature and pressure are stable over a long period, the pocket of oil is not submitted during this period to turbulances, allowing the formation of phospholipids in the oil phase vesicles. The oil-water interface between the two main phases will be, in this case, constituted with the duration of phospholipids from vesicles that migrated to the main water phase and having lost their water.
For a phospholipide vesicle can give a phospholipide bilayer, it requires that the lipid leaflet does not break and does not destroy the lipid leaflet forming the interface of the main phases. These vesicles will accumulate at the bottom of the oil phase. When the pressure of the vesicles that are above becomes large enough, the firsts in contact with the main sheet either will burst as we have described above or will detach to enter the main water phase surrounding himself with a piece of the main sheet, a length equivalent to the length of its inner leaflet.
This scenario of the formation of liposomes gives two homogeneous layers but structured differently with its inner leaflet and the water of the cytoplasm specific to each liposome.
Hypotheses on the molecular evolution from prebiotic processes to biotic metabolism.
[edit | edit source]Introduction
[edit | edit source]- It should be clear again that we start with a reaction medium for many kinds of small molecules, forming a liquid in thermodynamic equilibrium and surrounded by a surface ionized, and we do not start with a colloidal gel concentrated with macromolecules bearing large dynamic electromagnetic fields related to their large surface area with numerous positive and negative charges at a time.
The starting point of the prebiotic process of the pocket of abiotic oil is a liquid mixture in contact with the wall negatively ionized by carboxylic heads of fatty acids and neutralized with a mixture of cations. This set should evolve towards the state of a cell composed of a colloidal gel surrounded by a membrane negatively ionized and neutralized by almost only K+ cations. It is interesting to note that discoveries in research on the origins of life, on trapping organic molecules (including P) by mineral surfaces, involve surfaces positively ionized and neutralized by anions Cl-, the most cases.
In what follows I favored the sequestration process of aa and other molecules in addition to fixing the hydrophilic heads. There are other processes for selecting aa for molecular evolution. Sequestration appears as a rude sort of the head of aa (two closer functions, amine and carboxylic acid) then their limit size. When I suggested to the sequestration of aa, I had extended my thinking to imagine their potential and some have raised interesting issues with respect to this hypothesis.
There is no question of not submitting the aa selection to only a single process of sequestration, as the characteristics for the selection of 20 aa are many. We can ask many questions as, why this number almost immutable and why these aa specially?
- I found five processes that can determine the form of aa and the chemical function of their radicals:
- Diffusion through the membrane (see prebiotic chemo-osmosis).
- Sequestration by the mechanical cohesion of the liposome.
- Aminoacylation of the phosphate of membranes for fixing proteins to these membranes.
- Constraints of the metabolic pathways of synthesis. Syntheses that can take place also in the water away from the wall ...
- The chemical functions to be performed, including:
- - Nesting in the hydrophobic part of the lipid bilayer (hydrophobics aa) and general participation in the 3 D structure of proteins interacting with other proteins and membranes by electrical attraction and hydrogen bonds between the electric dipoles (polars and ionizables aa);
- - Action at a distance by electromagnetic fields (aromatics aa) and
- - Participation in chemical reactions themselves, ie creation and deletion of covalent bonds (reactifs aa).
In what follows we will:
- Illustrate the strategy implemented by the living to prepare the hydrophilic heads: Beyond the chirality ago in general spatial conformation, which explains the involvement of CTP and dCTP nucleotides for the establishment of the mobile arm.
- Study the case of chirality and homochirality of sugars whose origins are in the mechanical cohesion of the liposomes: the hydrogenation of L-glyceraldehyde-3P to Glycerol-3P on the fixed arm descriminate between L and D-glyceraldehyde.
- And implement the mechanical cohesion of the liposome for initialization of prebiotic metabolism: Here we study the initialization and the stages of the evolution of prebiotic metabolism by considering in addition to sequestration of aa, the dCTP and three coenzymes. These coenzymes intervene in the following order: prebiotics aa, B6, dCTP, B1, CTP and THF.
Beyond the chirality ago the spatial conformation over all.
[edit | edit source]
- For notation like enzymes (EC 4.3.1.7; 260-1-0), see Figure 1 and 2 below.
- In the following chapters I added the following abbreviations, because repetitive:
- GA, DGA and LGA for glyceraldehyde racemic, D and L;
- DHA for dihydroxyacetone;
- PE, PC, PS for phosphatidylethanolamine, phosphatidylcholine, phosphatidylserine.
- PtdGro, Ptd2Gro for phosphatidylglycerol and diphosphatidylglycerol (or cardiolipin).
- Search for enzyme in KEGG data base, clic here: [1]
- Search for enzyme in KEGG data base, clic here: [1]
- For notation like enzymes (EC 4.3.1.7; 260-1-0), see Figure 1 and 2 below.
- The chirality of the glycerol-P is set for:
- - The hydrogenation of the glycérone-P, achiral molecule that promote tautomerism;
- - The phosphorylation of glycerol, achiral molecule by symmetry, for attaching the phosphate after positionning the glycerol.
- The binding of serine can position its NH 2, as seen in the drawing 1-RG. It is the only movable arm carrying an extremely reactive function, the carboxylic function.
- The binding of glycerol as a movable arm, admirably illustrates the need to bypass the selection of the proper conformation among a multitude of other conformations. This strategy is also used for the creation of the ethanolamine arm after fixation of serine, as we shall see in what follows.
- The creation of the PE does not go through the attachment of free ethanolamine. In addition to the positioning of NH2 by serine, PE illustrates the importance of the spatial conformation in general by the fact that ethanolamine has no asymmetric carbon, but also by the need to eliminate all barriers to the overall mechanical cohesion of the liposome. Because the free ethanolamine, a very small molecule, may be beyond the control of macromolecules and poorly positioned or even establish hydrogen bonds with the fatty acids destroying the overall cohesion.
- The liposome, indeed, is not rigid. All PLD molecules of the bilayer are in constant motion while ensuring the cohesion of the whole. In prokaryotes, indeed, ethanolamine occurs only in a single reaction (EC 4.3.1.7; 260-1-0), which destroys it to acetaldehyde and NH3. In eukaryotes it is marked by a larger molecule that is the CDP (EC 2.7.1.82, 2.7.7.14;). Which prevents it from becoming a movable arm positioned incorrectly. Eukaryotes other than fungi (EC 2.7.8.8; 860-70), the PS is synthesized from the PE and serine (EC 2.7.8.29; 0-0-40).
- It is remarkable to note that the free choline, with his big head of trimethylamine (EC 2.7.8.24; 100-0-0), no tag is used directly to synthesize the PC in many prokaryotes but in any eukaryote, such as serine does with the PS in all prokaryotes and fungi but not in higher eukaryotes. Choline also differs yet ethanolamine in that it is not destroyed immediately, but it is degraded in several steps to glycine.
- But in all living synthesis of the movable arm passes through an intermediate unexpected esterification by CTP. This is an essential difference with the theory of prebiotic metabolism based on the direct esterification of small molecules. And this intermediate step includes all basic biotic metabolic namely a nucleotide (CTP), a désoxynuléotide (dCTP) with a ratio CTP/dCTP equal to 0.88 (in Biochemistry of lipids, lipoproteins and membranes 2008, page 74 [8]), a phospholipid and of course an enzyme. But does not involve coenzymes, CoA or B6. Here we have gathered into monomers, DNA, RNA, protein and membrane. We will see in what follows that this esterification step has a sole purpose is to implement properly the movable arm so that it can fulfill its primary function, the mechanical cohesion of the liposome.
-
Figure 1. This diagram represents the compilation of organisms with a given gene in the pathway of glycerophospholipids: According to a screenshot of the database of metabolic pathways of KEGG. The colored rectangles correspond to membrane enzymes. KEGG.
The colored rectangles correspond to membrane enzymes. The compilation is a rough and personnel count of organisms listed in the database, and consists of two numbers, the first that of prokaryotes and the second that of eukaryotes.
When the number is weak, second count is do from lists of Brenda or RefSeq databases (from the links provided by KEGG) and is coupled with that from the list of KEGG. Example: 0.5.60 corresponds to 0 prokaryotes in the list of KEGG, 5 prokaryotes in Brenda and RefSeq, and 60 eukaryotes in KEGG. The drawings of arrows and the names of molecules are those of KEGG. -
Figure 2. Analogous to Figure 1, according to the KEGG metabolic pathways. Here is a montage from several metabolic pathways to represent traffic between Serine, Glycerone, fatty acids and phospholipids:Fatty acid Biosynthesis, Fatty acid Metabolism, Glycerolipid Metabolism, Glycerophospholipid Metabolism et Glycolysis.
The mechanical cohesion of the liposome is at the origin of chirality and homo-chirality of sugars.
[edit | edit source] In biotic metabolism all sugars have the D configuration for the first asymmetric carbon following the aldehyde or ketone. This is particularly the case of D-glyceraldehyde at the base of the synthesis of all biotics sugars. But in the theory of the mechanical cohesion of the liposome we have shown that the homochirality of glycerol-3P, serine and possibly other amino acids if their sequestration by liposomes is effective. In addition, the DGA is not part of the lipid bilayer and acts only second to the synthesis of glycerol-3P.
How then has established homochirality of DGA? Can we involve the mechanical cohesion of the liposome? If this were the case, would have to be sequestered as aa, ie take the place of the mobile arm of the hydrophilic head and its chirality must be L. So the LGA could have been sequestered and benefited from the DGA. How then did the LGA could completely disappear in molecular evolution to make room for DGA alone? Once attached the LGA can it be hydrogenated glycerol? But it must be assumed one hydrogenation different from that the fixed arm.
- The disappearance of the LGA from the biotic metabolism is due to its attachment to the fixed arm in competition with DHA.
- We saw in chapter Prebiotic glycerol-phosphate chirality, for bacteria, that the glycerol-P fixing the fatty acids is an L-glycerol-3P. It has the same conformation as the LGA-3P with an aldehyde instead of alcohol terminal. So the LGA can take the place of DHA. Then there for hydrogenation to obtain glycerol-3P with the tautomerism of LGA (as was supposed to glycerone) to facilitate and enable, as a first step, the appropriate spatial configuration.
- So the elimination of LGA leads, if not its total disappearance (depending on the degree of reversibility of the isomerization of prebiotic GA: L <-> D ), at least its selective disadvantage compared to the DGA that will remain free and unattached to PLD. Then the homochirality of sugars can begin. Thus we see that the homochirality of aa stems from their sequestration or their attachment (serine) in the PLD free arm and is still in the biotic metabolism, while the homochirality of sugars had to be done only once during prebiotic metabolism, and the LGA disappears completely after.
- PtdGro chirality
- Glycerol-P PtdGro of the mobile arm is L-glycerol-3P. Indeed it has the same configuration as L-serine with terminal alcohol (CH2OH) outside of the membrane. This is the same as on the fixed arm since it has terminal alcohol disposed inwardly of the membrane (see chapiter Prebiotic glycerol-phosphate chirality). PtdGro is declined, in Kegg, in phosphatidylglycerol without specifying the chirality of the glycerol. In my opinion it should be called phosphatidyl-L-glycerol (without adding 3P which would be confusing). Moreover, the reaction is clearly written in Kegg [9]:
- CDP-diacylglycerol + sn-glycerol 3-phosphate = CMP + 3(3-sn-phosphatidyl)-sn-glycerol 1-phosphate (EC 2.7.8.5; 1200-110)
- P of the sn-glycerol 1-P refers to the P terminal, while the substrate is sn-glycerol 3-P .
- CDP-diacylglycerol + sn-glycerol 3-phosphate = CMP + 3(3-sn-phosphatidyl)-sn-glycerol 1-phosphate (EC 2.7.8.5; 1200-110)
- The PtdGro also exists in the hydrophilic heads of the Archaea.[10]. We have seen that in bacteria, the L-glycerol-3P could come from the hydrogenation of DHA or LGA-3P. For archaea, at early prebiotic evolution, if the D-glycerol-3P had coming from DGA-3P, someone would have had a homochirality L of all sugars. This is not the case. So archaea, with all their sugars in D chirality form, appeared after the bacteria.
- It should be noted however that the synthesis of the D-glycérol-3P and its use are still possible even in the biotic metabolism. Indeed the Ptd2Gro (EC 278. -, Cls, 700-100), ubiquitous, has a glycerol flanked by two phosphatidic acids with 4 hydrolysable ester bonds. The release of glycerol-P can be done in two possible ways each giving a glycerol-1P or a glycerol-3P (in Biochemistry of lipids, lipoproteins and membranes 2008, page 75) [8].
- One would say that the first movable arms that would have occurred, were of L-glycerol-3P since these have already been synthesized in the first step of the formation of the hydrophilic head. But it should be noted that the synthesis of PtdGro request a step further than the PS from a glycerol that must be detached from the fixed arm; as glycerol, if is the single movable arm, may establish under certain conditions of temperature and pressure generalized hydrogen bonds, since it is not ionizable, which crystallize the liposome. We then see the importance of serine that can be synthesized abiotically [5], even in small quantities, or it can be synthesized in a single enzymatic reaction (in the presence of B6), in biotic metabolism (EC 431.17, 800-20, and 431.19, 930-100). Serine is interesting because it is a reactive amino acid in its zwitterionic head and in one step can give PE which the amine is more reactive than glycerol.
CMP positions the phosphate tetrahedron to esterify the free arm in the proper configuration necessary for the mechanical cohesion of the liposome.
[edit | edit source]- We saw in paragraph spatial conformation, the essential difference between unexpected and hypothetical prebiotic esterifications and biotic esterifications. Considering the spatial configuration in general instead of one chirality, it is clear that in prebiotic, the number of configurations explodes when considering the rotation of the free phosphate before its esterification by serine, glycerol, ethanolamine or choline.
- In fixing the glycerol of mobile arm in biotic metabolism, phosphate (of glycerol-3P) was used for the tag and perhaps forcing it to be fixed in the correct position of the mechanical cohesion. But the P of PtdGro-P make heavy the movable arm (EC 2.7.8.5; 1200-110). Phosphate is quickly expelled in the reaction EC 3.1.3.27 (500-4-0) in most prokaryotes.
- There are three enzymes that position phosphate (Ec 2.7.7.41, 1200-140) for serine and glycerol, (Ec 2.7.7.14, 0-0-20) for ethanolamine and (Ec 2.7.7.15, 25 -70) for choline. We see that only the first is generalized to all living beings, while two others are more specific to eukaryotes only.
The mechanical cohesion of the liposome and the initialzation of prebiotic metabolism.
[edit | edit source] Let us go back to the conditions of the prebiotic pocket of oil as has been described earlier, and reconsider the initialization of prebiotic metabolism in the light of results achieved to date in a aqueous vesicle of phase oil.
We had assumed that the initialization of metabolism could start to conditions as necessary molecules could be trapped by the wall of the vesicle, displacing regularly the thermodynamic equilibria of aqueous contents. Except for the hydrogenation of DHA or GA by H2, hydrogenation of which it was assumed the existence, all other reactions for the synthesis of hydrophilic heads are esterifications, very slow when there are no enzymes. These are in order:
- - Phosphorylation of DHA and LGA;
- - Esterification of these molecules phosphorylated with fatty acids after in-situ hydrogenation, catalyzed by phosphate and carboxylic surface.
- - Fixing the ethanolamine or serine or glycerol. Assuming that this latter can exist, for example by its hydrolysis from the wall.
- The results of the inquiry into the theory of mechanical cohesion of the liposome shows that it creates a virtuous circle: the increase of hydrophilic heads of the same chirality increases the cohesion of the vesicle, which in turn promotes the binding of molecules of the same chirality, forming new hydrophilic heads.
- This virtuous circle of mechanical cohesion will also create another virtuous cycle based on catalysis: L-serine hydrophilic heads has its carboxyl free and very reactive; the mechanical cohesion also concentrating more and more L-aa on a large surface with more and more hydrophilic heads, favoring the grouping of amino acids in catalytic entities, in increasingly effective.
- Some studies (Wieczorek, 2009 [11]) showed for example that the Ser-His dipeptide, attached to the wall, is very reactive and is responsible for many processes occurring in the liposome. It is found in the active sites of many enzymes. In the case of prebiotic metabolism, where reactions are very slow, a single molecule of this dipeptide may have a strong acceleration of molecular evolution.
- Sequestration of some coenzymes.
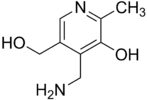
- The sequestration process is really interesting for some precursors of coenzymes (see their pictures below) as pyridoxamine for pyridoxal-P, the pyrimidine ring of thiamine, the pteridinic ring of folic acid, and deoxycytidine. These are small molecules of the size of aa with amine and hydroxyl as ethanolamine. In addition deoxycytidine is fixed directly in the form of dCMP on the PLD such as serine in the biotic metabolism.
- The analogy of the three other coenzymes with dCTP is even more striking because their intermediates are phosphorylated before being converted into their active form, as deoxycytidine: this is how the pyrimidinic ring of thiamine (CBS 271.49, 2747) and the pteridinic ring of folic acid (EC 2763) are phosphorylated with two phosphate in biotic metabolism. Curiously pyridoxamine, when she has, is converted into the active form, pyridoxal-5P, in Clostridium kainantoi by a transaminase (EC 261.54) from D-Ala or D-Glu. Transaminase of this type is normally done with an L-aa and as a coenzyme pyridoxal-5P.
- We will assume that in the prebiotic metabolism, these precursors are first sequestered (or fixed?), On the wall before being converted into their active form. Particular we will see (Table 1, step 5) that the dCTP, essential for biotic metabolism, might be interesting to introduce it before the onset of CTP since this latter requires ATP for its synthesis (step 6), while the dCTP might appear before (step 4).
-
4-Amino-5-hydroxymethyl-2-methylpyrimidine
-
Thiamine
-
4-Methyl-5-(2-hydroxyethyl)-thiazole
-
desoxyCytidine
-
Acide folique
-
2-Amino-4-hydroxy-6-hydroxymethyl-7,8-dihydropteridine
-
Acide 4-Aminobenzoïque
-
Pyridoxamine
-
Pyridoxal
- The steps of initializing the prebiotic metabolism, Table 1:
We will now place the script initialization of prebiotic metabolism starting with the synthesis of hydrophilic heads generating mechanical cohesion and differentiation between the wall rich in sequestered L-aa and the liquid inside rich in D-aa.
Then comes the sequestration of B6 which would act as a coenzyme for groups of L-aa of the wall. I introduced the first in B6 by analogy, because in the biotic metabolism, activates countless reactions between aa, and between aa and NH3. Thereby increasing tenfold the virtuous cycle of catalysis.
Table 1. Mechanical cohesion and the steps of initializing the prebiotic metabolism.
| Fixation | Sequestered | Products | Accumulation | Removal | Utilization | aa present | Comments from the KEGG website | ||
|---|---|---|---|---|---|---|---|---|---|
| (analogy enzymes with groups of L-aa sequestered). | |||||||||
| 0 | Vesicle. Prebiotic Energetics : Formose. Hydrothermale Synthesis of aa : ADEGS. | ||||||||
| (for analogy with the synthesis of L-aa see the KEGG website). | |||||||||
| 1 | DHA LGA L-ser | Hydrophilic heads PS | DGA D-ser cohesion | LGA | H2 | ADEGS | prebiotic catalytic Hydrogenation, without enzymes, of DHA and LGA and not of DGA. Accumulation of D-ser and homochirality of sugars is due to fixing the LGA. | ||
| 2 | L-aa | groupes L-aa | D-aa | initial free L-ser | ADEGS | ADEGS | Differenciation between the surface (L-aa) and the aquous inside (D-aa). | ||
| 3 | B6 | L-ser NQCTWY ADEG PE PtdGro Ptd2Gro |
cohesion 2-oxo-acids |
initial free aa | H2S NH3 indol phenol acetaldehyde | NQCTWY ADEGS | B6 et homogeneous grouping of amino acids catalyze better the reactions of 1 step without ATP, using NH3. Interconversions between aa accumulate oxo-acids that with DHA and DGA, they prepare the intermediate metabolism of carboxylic acids. | ||
| 4 | dR-1P dATP | DGA-3P+acetal Adenine | Reactions without coenzymes : 4124, 5427 produce D-dRibose-1P. then with Adenine : 2421,271.76,2743,2746 produce dATP. | ||||||
| 5 | CTP | cytosine | cytosine+dR-1P | Cohesion | Cytosine dR-1P dATP | equivalent of 2421 do not exist for cytosine ; groups of L-aa could catalyze the synthesis of dCTP in-situ. | |||
| 6 | B1 | ATP CTP NAD B6 SAM FAD FMN Biotine FHKPIVM PC |
Cohesion | Initial H2 and B6 | Bases nicotinate DHA+DGA dATP | FHKPIVM NQCTWY ADEGS | B1 consists of m1 and m2, m1 can be sequestered. The synthesis of B1 can be done in-situ as for dCTP. With B1, 412.13, 313.11, 2211, 5131, 5316 and from DHA+DGA is obtained R-5P that with dATP can done PRPP (2761) and R-1P (5427). A+R-1P+PRPP+PPP → ATP : 2421, 2428, 2743, 2741. C+R-1P+ATP → CTP : 2422, 271.48, 274.14, 2746. N+PRPP+ATP+NH3 → NAD : 242.11, 2771, 6351. D-Ribulose-5P (5131)+DGA-3P+L-gln → Pyridoxal-P (B6) : 4.-.-.-, YaaD, Pyridoxal biosynthesis lyase pdxS. 2.6.-.-, YaaE, Glutamine amidotransferase subunit pdxT. | ||
| 7 | THF | CoA fatty acids LR | DHA and DGA from formose, Phosphate | LR FHKPIVM NQCTWY ADEGS | THF consists of m3 and m4 that can be sequestered. Pyruvate+B1+NAD+THF+ATP+L-asp+L-cys → CoA : 2216, 11.86, 4219, 212.11, 111.169, 411.11, 6321, 271.33, 6325, 411.36, 2773, 271.24. | ||||
| . | |||||||||
| 8 | Liposomes formation : getting through membrane energetics. | ||||||||
| . | |||||||||
| 9 | DHA DGA Bases nicotinate indol phenol | Initial bases nicotinate indol phenol | Present metabolisme | Present L-aa | See KEGG website | ||||
| 10 | B1 THF | Initial B1 THF | Present metabolisme | Present L-aa | See KEGG website | ||||
- Step 0.
- These are the starting materials in the aqueous vesicle in the oil phase. Every molecule is susceptible to be present. However for concentrations I refer to experiments at high temperature (150 ° C) and high pressures (300 bar).
- - H2 H2S CO2 N2, then NH3 : gases of hydrothermal vents (Charlou [12] 2002, Proskurowski [13] 2008 ).
- - Phosphates and polyphosphates of seabed (Arrhenius 1997 [14]).
- - alkanes, fatty acids, alcohols and aldehydes from Fischer-Tropsch process which acetaldehyde (Rushdi[15] 2001, McCollom 1999,[16] 2006 [17]).
- - dihydroxyacetone glyceraldehyde glyoxal by hydroformylation or formose reaction at 120 bars and 140°C [4].
- - amino acids ADEGS produced in hydrothermales experiments with nitrogene molecules [5].
- - Precursors of coenzymes and aromatic rings in very small amounts of prebiotic pocket of oil (hypothesis): nucleic bases, AGCUT; pyridoxamine, the two nuclei of thiamine, the two nuclei of folate and nicotinate for NAD.
- Step 1.
- Synthesis of hydrophilic head following the analysis of the previous paragraphs. Fixing on the wall of L-ser, DHA and LGA, and accumulation of D-ser DGA in water. Origin of homochirality of sugars via DGA. Virtuous cycle (synthesis of heads) / (mechanical cohesion).
- Step 2.
- - Sequestration of L-aa by the wall and concentration of D-aa in water. L-ser is gradually disappearing from the water with the decrease in the hydrothermal synthesis of aa with time.
- - Combination of L-aa on the wall to form pseudo-enzymes. They are perhaps not very effective, but multiple combinations, more or less ephemeral, are possible. Over the surface of hydrophilic heads grew more groups could be strong and numerous, they will be more cooperative in catalysis and in their grouping itself. This is the virtuous cycle of catalysis subtended by the mechanical cohesion.
- Step 3.
- - The accumulation of DGA and D-ser displaces very slowly the equilibriums towards L-ser.
- - Sequestration of B6 accelerates the isomerization of D-ser to L-ser, the deamination of D-ser to pyruvate (Ec 431.18) and its amination to L-ser (CBS 431.17).
- - Under the action of B6, synthesis of new aa: From pyruvate, NH3 and indole (Trp) or phenol (Tyr). Thiolysis of hydrophilic heads with H2S (Cys). Condensation of Gly and acetaldehyde forming Thr. Amination of Glu to Gln and Asp to Asn (see KEGG for the analogy with enzymes).
- - Under the action of B6, decarboxylation of PS serine to give PE (EC 411.65). Ethanolamine produced very little in the hydrothermal synthesis of aa, has been fixed in place of serine but much more difficultly as we have seen previously. There are mechanical cohesion strengthening by PE because it has no reactive heads.
- - Under the action of B6, Thr deamination to give 2-oxo-butanoate (EC 4125) further required for the synthesis of Ile Val Leu.
- - Transaminations between aa (ADEGS more new CNQTWY) for the production of L-Ser, which accumulates 2-oxo-acids.
- - DGA, DHA, 2-oxo-acids, NH3, H2S, ADEGS and CNQTWY are prebiotic intermediary metabolism.
- - All these new products through B6 are obtained in a single reaction.
- Step 4.
- - Synthesis, without coenzyme, of the first deoxy-pentose in two reactions (EC 4124, 5427) which the second is autocatalytic (formation of ribose bisphosphate): DGA-3P + acetaldehyde = D-dRibose-1P.
- - Synthesis of deoxy-adenosine without coenzyme (EC 2421), then 2 phosphorylation with P and PPP give the dADP (EC 3135, 2743).
- - The passage to dATP require ATP in the biotic metabolism (EC 2746). I assume, in prebiotic metabolism, that is entirely possible that dATP is formed very slowly by self-catalysis (confusing dATP and ATP in EC 2746) or in the presence of polyphosphates such as two reactions that precede it. Besides, this is an esterification and I founded my theory of prebiotic molecular evolution on esterification (see Section 5.1.2).
- Step 5.
- - This step may seem theoretically superfluous , but step 6 below requires thiamine and 7 reactions to arrive at the ribose-1P. Now we have seen that the sequestration of thiamin, folate and dCTP need an in-situ condensation of two parts to form each coenzyme. The dCTP should appear before the CTP.
- - In the biotic metabolism, there is no equivalent of (EC 2421) for dCTP as in step 4. Now the passage by the CTP or dCTP for fixing the movable arm (EC 277.41), regardless of the arms, seem crucial. Also the formation in situ of dCTP in the presence of groups of sequestered L-aa, rapidly bring high mechanical cohesion. The dCTP acts as a coenzyme since dCTP, after hydrolysis can be regenerated by phosphorylation of dCMP.
- Step 6.
- - In-situ synthesis of B1 (see drawing molecules below).
- - It is the accumulation of DGA in the first stage of the scenario that will promote, by displacement of thermodynamic equilibrium, its condensation with DHA, both in phosphorylated form: DGA + DHA-3P-P (EC 412.13).
- - ATP: There is six reactions and the participation of B1 and dATP, only coenzymes, to arrive at the central molecule PRPP then isomerization to reach the Ribose-1P required for the synthesis of ATP from the adenine and PPP.
- - The synthesis of CTP requires ATP.
- - The synthesis of NAD requires ATP and nicotinic acid.
- - The synthesis of S-adenosylmethionine (SAM), flavins (FAD and FMN) and biotin requires only prebiotics coenzymes already created.
- - The pyridoxal-5P (B6) begins with an intermediate in the synthesis of D-ribose-1P, D-ribulose-5P: D-ribulose-5P-3P + DGA + L-Gln (EC 5131).
- - Cohesion will develop at high speed with the CTP. Replacing the prebiotic hydrogenation (H2) by the hydrogenation by the NAD and B6 is synthesized de novo.
- - 7 new aa can be synthesized which histidine we have seen, important for the active sites of proteins: FHKPIVM.
- - The synthesis of methionine allows the synthesis of SAM and hence the production of PC.
- Step 7.
- - In-situ synthesis of folate (THF) (see drawing molecules below).
- - Synthesis of coenzyme A, the last 2 aa LR and fatty acids.
- Step 8.
- - The formation of the liposome with the passage of the vesicle in the water phase, as we have seen, is necessary because at one time or another, limited energy into prebiotic vesicle (formose: DHA and DGA), will be exhausted. The prebiotic energy will be replaced by the membrane energy. The membrane follows molecular evolution assumed in the article of prebiotic chemo-osmosis.
- - The basic molecules of metabolism, including phosphate, then arrive by diffusion first, then by physical flip-flop for phosphate, by primitive porins, equivalent to ionophores, and finally by the carrier protein.
- - Energy protein complexes take hold into the membrane by amino acids migration from inside or by diffusion of those from outside.
- Step 9.
- - With membrane metabolism, synthesis of DHA and DGA de novo, synthesis of fatty acids and coenzymes such as membrane cytochromes. Replacement of the aromatic rings: indol, phenol, nicotinate and nucleic bases.
- Step 10.
- - Rplacement of thiamine and folic acid (THF).
I stop here the molecular evolution of metabolism. Another thought would be interested in full to the evolution of macromolecules from this metabolism that would provide the monomers.
References
[edit | edit source]- ↑ http://link.springer.com/article/10.1007/s11084-015-9416-7?sa_campaign=email/event/articleAuthor/onlineFirst
- ↑ http://www.genome.jp/dbget-bin/www_bget?C00093
- ↑ Dorota Wolicka, Andrzej Borkowski, and Dariusz Dobrzynski: Interactions between Microorganisms, Crude Oil and Formation Waters. Geomicrobiology Journal, 27:43–52, 2010.
- ↑ 4.0 4.1 Zoltán Györgydeák,István F. Pelyvás. Monosaccharide sugars: chemical synthesis by chain elongation, degradation...(Page 8). Academic Press 1998.
- ↑ 5.0 5.1 5.2 A. D. Aubrey & H. J. Cleaves & Jeffrey L. Bada: The Role of Submarine Hydrothermal Systems in the Synthesis of Amino Acids. Orig Life Evol Biosph (2009) 39:91–108 DOI 10.1007/s11084-008-9153-2
- ↑ http://www.ifremer.fr/serpentine/fiches/fiche8.htm
- ↑ Daniel Segré and Doron Lancet: Composing life. EMBO Reports vol.1 no.3, pp 217–222, 2000. http://ool.weizmann.ac.il/Segre_Lancet_EMBOrep_2000.pdf
- ↑ 8.0 8.1 Biochemistry of lipids, lipoproteins and membranes 5th edition, 2008: edited by D.E. Vance and J.E. Vance; Elsevier
- ↑ http://www.genome.jp/dbget-bin/www_bget?ec:2.7.8.5
- ↑ Hiromi Daiyasu et al. : A study of archaeal enzymes involved in polar lipid synthesis linking amino acid sequence information, genomic contexts and lipid composition. Archaea 1, 399–410 © 2005 Heron Publishing—Victoria, Canada
- ↑ Gorlero M, Wieczorek R, Adamala K, Giorgi A, Schininà ME, Stano P, Luisi PL. (2009) Ser-His catalyses the formation of peptides and PNAs. FEBS Lett. 583(1):153-6.
- ↑ Charlou J.L., Donval J.P., Fouquet Y., Jean-Baptiste P., Holm N., « Geochemistry of high H2 and CH4 vent fluids issuing from ultramafic rocks at the Rainbow hydrothermal field », Chemical Geology, vol. 191, 2002, p. 345-359. sciencedirect
- ↑ Giora Proskurowski, Marvin D. Lilley, Jeffery S. Seewald, Gretchen L. Früh-Green, Eric J. Olson,1 John E. Lupton, Sean P. Sylva, Deborah S. Kelley: Abiogenic Hydrocarbon Production at Lost City Hydrothermal Field . Science vol 319, 1 février 2008 sciencemag
- ↑ G. Arrhenius, B. Sales, S. Mojzsis and T. Lee : Entropy and Charge in Molecular Evolution-the Case of Phosphate Journal of Theoretical Biology Volume 187, Issue 4, 21 August 1997, Pages 503-522 sciencedirect
- ↑ Ahmed I. Rushdi, and Bernd R.T. Simoneit: Lipid formation by aqueous fischer-tropsch-type synthesis over a temperature range of 100 to 400 Template:Abréviation. Origins of Life and Evolution of Biospheres (2001) 31: 103–118.
- ↑ T.M.McCollom et al. 1999: Lipid synthesis under hydrothermal conditions by fischer-tropsch-type reactions . Origins of Life and Evolution of the Biosphere 29: 153–166, 1999
- ↑ T.M.McCollom et al. 2006:Carbon isotope composition of organic compounds produced by abiotic synthesis under hydrothermal conditions. Earth and Planetary Science Letters Volume 243, Issues 1-2, 15 March 2006, Pages 74-84