Enzyme structure and function
| Subject classification: this is a chemistry resource. |
| Type classification: this is a lesson resource. |
In this lesson, the three-dimensional structure of proteins will be discussed: the primary structure of polypeptides, secondary structures in proteins (α-helix, β-sheet), and the tertiary structure. The concept of an enzyme active site will be introduced.
Aims
By the end of this lesson, you should
- be familiar with the main features of protein structure
- understand the concept of an enzyme active site
What is an enzyme?
[edit | edit source]
- Enzymes are protein macromolecules. With the exception of catalytic DNA and RNA which are considered enzymes but not proteins.
- They have a defined amino acid sequence, and are typically 100-500 amino acids long.
- They have a defined three-dimensional structure.
- Enzymes are catalysts.
- They act as a catalyst to a chemical or biochemical reaction, with a defined mechanism.
- They increase the speed of that reaction, typically by 106-1014 times faster than the rate of the uncatalysed reaction.
- They are selective for a single substrate.
- They speed up rate of reaction by lowering the activation energy (Ea).
- They are stereospecific, meaning the reaction produces a single product.
Structure of Enzymes
[edit | edit source]Primary structure
[edit | edit source]Enzymes are made up of amino acids which are linked together via amide (peptide) bonds in a linear chain. This is the primary structure. The resulting amino acid chain is called a polypeptide or protein. The specific order of amino acid in the protein is encoded by the DNA sequence of the corresponding gene.
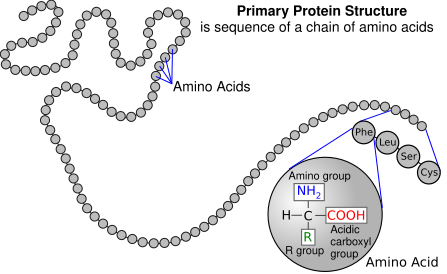
Click here for a list of all 20 amino acids.
Secondary structure
[edit | edit source]The hydrogen in the amino group (NH2) and the oxygen in the carboxyl group (COOH) of each amino acid can bond with each other by means of hydrogen bond, this means that the amino acids in the same chain can interact with each other. As a result, the protein chain can fold up on itself, and it can fold up in two ways, resulting in two secondary structures: it can either wrap round forming the α-helix, or it can fold on top of itself forming the β-sheet.
-
α-helix
-
β-sheet
-
Molecular diagram of β-sheets
In the images above, the dotted lines represent the hydrogen bonds. There are two forms of β-sheet, depending on the direction of the protein chain. If the direction alternates between every fold, it forms an anti-parallel sheet; if it remains the same direction, it forms a parallel sheet.
Tertiary structure
[edit | edit source]As a consequence of the folding-up of the 2D linear chain in the secondary structure, the protein can fold up further and in doing so gains a three-dimensional structure. This is its tertiary structure.

Substrate binding
[edit | edit source]All enzymes have an active site, where the reaction is catalysed. This part of the enzyme has the specific shape and functional groups to bind to the reacting molecules (called the substrate). Hence the active site contains a small number of catalytic amino acids, which are essential in catalysing the reaction. The substrate molecule can bind to the active site via non-covalent interactions:
- electrostatic interactions
- for example, those amino acids in section A of this list will attract an oppositely charged substrate.
- hydrogen bonding
- typically between the amide and carboxyl groups of the amino acid can form hydrogen bonds with the substrate.
- Van der Waals interactions
- for example, those amino acids in section B of this list.
- hydrophobic interactions
- for example, those amino acids in section D of this list will repel water.
![]() Next lesson: Enzyme catalysis
Next lesson: Enzyme catalysis



